
Introduction
Flow assurance challenges in oil and gas operations are often associated with mineral scale, hydrates, or corrosion. Yet in many crude oil production systems, especially mature fields, the most persistent threat to uninterrupted flow is organic deposition — specifically wax accumulation. Unlike sudden mechanical failures, wax deposition is gradual and often underestimated. It builds silently along tubing walls and pipelines, narrowing flow paths, increasing pressure drop, and reducing production efficiency.
Paraffinic crude oils contain long-chain hydrocarbons that remain dissolved under reservoir conditions. However, as these fluids travel from the high-temperature reservoir to cooler surface facilities, the delicate thermodynamic balance shifts. Once temperature drops below a critical threshold, wax molecules begin to crystallize and deposit on metal surfaces. Over time, these deposits can restrict flow, overload artificial lift systems, and even cause complete blockages.
Industry data suggests that wax-related flow assurance issues account for significant operational expenditure in paraffinic fields worldwide. Cleaning operations, pigging schedules, solvent treatments, and production shutdowns collectively contribute to rising lifecycle costs. In offshore and subsea systems where intervention is complex and expensive, wax deposition becomes not just a maintenance issue but a strategic operational risk.
This article examines the fundamental mechanisms behind wax deposition and explores chemical strategies that transform wax control from reactive intervention to proactive flow assurance management.
Understanding the Chemistry of Wax in Crude Oil
Crude oil is a complex mixture of hydrocarbons ranging from light gases to heavy asphaltenes. Paraffin wax consists primarily of long-chain n-alkanes, typically containing 18 to 60 carbon atoms. Under reservoir temperature and pressure, these molecules remain dissolved in the liquid phase. However, solubility decreases sharply as temperature declines.
The critical parameter in wax behavior is the Wax Appearance Temperature (WAT). This is the temperature at which wax crystals first begin to form as the fluid cools. Below WAT, paraffin molecules start to organize into solid crystalline structures. These crystals grow, agglomerate, and eventually adhere to pipe walls.
Pressure also plays a role. As fluids ascend the wellbore, pressure reduction changes gas solubility and fluid composition. This can indirectly affect wax precipitation by altering temperature profiles and flow regimes. In long tiebacks and subsea pipelines, external seawater temperatures further accelerate cooling, making wax control especially critical.
The severity of wax deposition depends on several interacting factors: crude composition, cooling rate, flow velocity, pipe roughness, and shear conditions. Even small differences in crude characteristics can dramatically change deposition behavior. High-paraffin crudes are particularly vulnerable, but even moderate paraffin content can cause operational challenges when temperature gradients are steep.
Mechanisms of Wax Deposition in Production Systems
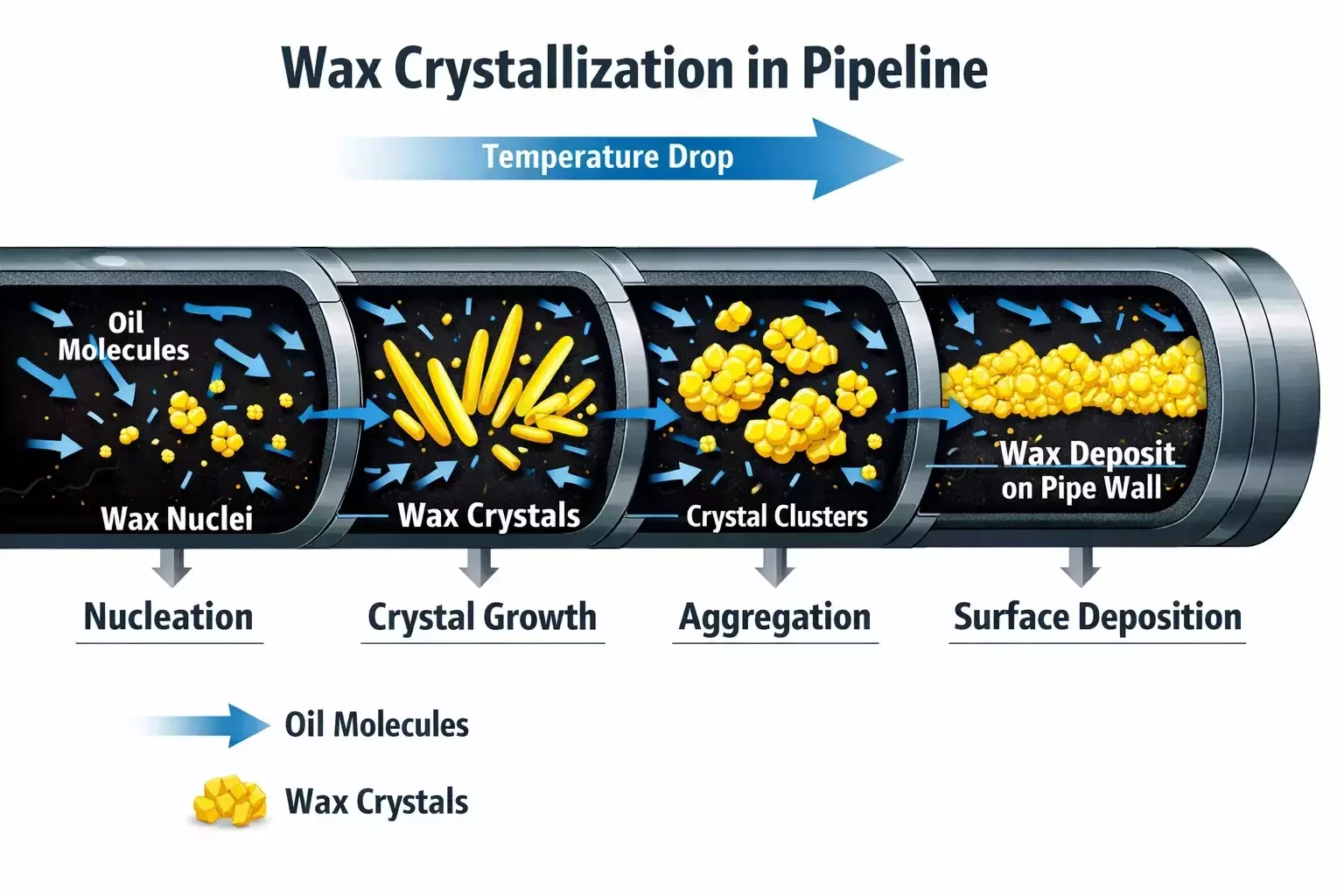
Wax deposition does not occur as a single-step event. It is a multi-stage process involving nucleation, crystal growth, agglomeration, and surface attachment.
The first stage is nucleation. As temperature falls below WAT, microscopic wax crystals form within the bulk fluid. These crystals are initially small and suspended. If flow velocity is sufficient, some crystals may remain dispersed without depositing.
The second stage is crystal growth. As cooling continues, additional paraffin molecules attach to existing nuclei, increasing crystal size. Larger crystals have a higher probability of interacting with pipe surfaces.
The third stage involves aggregation. Crystals collide and combine, forming clusters that increase in mass. These aggregates are more likely to settle in low-velocity zones, bends, or irregular surfaces.
Finally, deposition occurs when wax particles adhere to pipe walls. Surface roughness and existing deposits enhance attachment. Once a thin layer forms, it acts as an insulating barrier, accelerating further deposition by reducing heat transfer and maintaining lower wall temperatures.
Over time, the deposit thickness increases, effectively reducing internal diameter. This raises frictional pressure losses and forces pumps or compressors to work harder. In severe cases, flow becomes unstable or completely obstructed.
Operational Consequences of Uncontrolled Wax Deposition
The impact of wax accumulation extends beyond reduced flow rate. Increased pressure drop leads to higher energy consumption, raising operating expenses. Artificial lift systems such as electric submersible pumps experience higher load, shortening equipment life.
In pipelines, wax deposits create uneven flow profiles and can trap additional solids or debris. This increases the risk of slugging and mechanical stress. Pigging operations become more frequent, interrupting production and increasing intervention cost.
In subsea developments, wax deposition presents even greater challenges. Cold seabed temperatures accelerate crystallization, and intervention options are limited. A hydrate event combined with wax buildup can complicate restart procedures significantly.
From a financial perspective, wax-related downtime can cost operators substantial revenue per day in high-output fields. Preventive strategies are therefore not simply chemical decisions but economic safeguards.
Thermodynamics and Flow Dynamics Behind Wax Precipitation
To control wax effectively, it is essential to understand that deposition is fundamentally a thermodynamic imbalance. At reservoir conditions, paraffin molecules exist in equilibrium with the crude oil matrix. The moment the temperature begins to decline during production, the solubility curve shifts. Once the operating temperature crosses below the Wax Appearance Temperature, the system enters a supersaturated state.
Supersaturation is the driving force of crystallization. In this condition, the oil contains more dissolved paraffin than it can thermodynamically sustain. Nature responds by separating the excess into solid crystalline structures. The rate of precipitation depends on how rapidly the temperature drops. Rapid cooling can trigger intense nucleation, producing numerous small crystals. Gradual cooling, by contrast, favors slower crystal growth but may result in larger aggregates.
Flow velocity introduces another layer of complexity. High shear rates can delay deposition by keeping crystals suspended, but they cannot prevent nucleation. In laminar or low-velocity zones, especially in horizontal pipelines or dead legs, wax crystals settle and attach more easily. Turbulent flow reduces residence time at the pipe wall but may also increase crystal collision frequency, enhancing aggregation.
Heat transfer between the flowing crude and the pipe wall is equally critical. Metal surfaces exposed to colder external environments act as heat sinks. As the wall temperature drops below the bulk fluid temperature, a thermal gradient forms. This gradient encourages paraffin molecules to migrate toward the cooler surface, intensifying deposition along the wall even if the bulk fluid remains slightly above WAT.
Over time, the deposit layer itself becomes thermally insulating. This reduces heat exchange efficiency and maintains lower wall temperatures, creating a self-accelerating cycle of deposition. In long-distance pipelines, particularly subsea tiebacks, this effect becomes more pronounced with increasing length.
Understanding these thermodynamic and fluid dynamic interactions is central to designing effective chemical control strategies.
Chemical Strategies for Wax Control
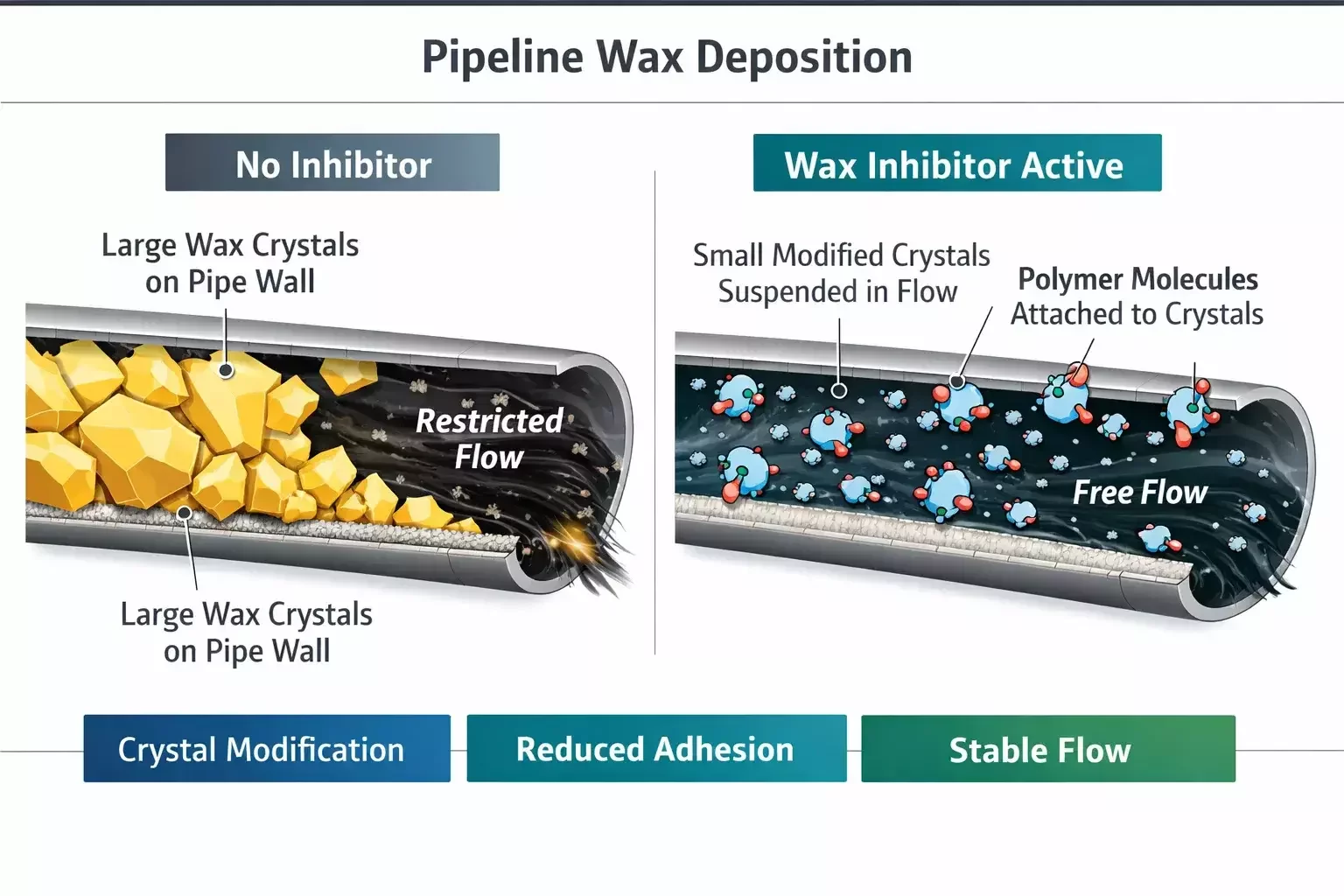
Mechanical cleaning methods such as pigging and hot oil circulation are widely used, but they address the symptom rather than the cause. Chemical treatment, when properly designed, modifies the crystallization process itself, reducing deposit formation at its origin.
Wax inhibitors, often referred to as crystal modifiers, function by interacting with paraffin molecules during nucleation and growth. These polymers adsorb onto the surface of forming crystals, altering their structure. Instead of developing into large, plate-like crystals that adhere to surfaces, the modified crystals remain smaller and more irregular in shape. This reduces their ability to interlock and attach to metal surfaces.
Pour point depressants operate through a similar mechanism but focus on lowering the temperature at which the crude loses flowability. By disrupting the formation of an interconnected crystal network, they allow the fluid to remain mobile at lower temperatures. While not always sufficient as a standalone solution, they are effective in combination treatment programs.
Wax dispersants take a different approach. Rather than modifying crystal growth, they keep precipitated wax particles suspended in the fluid. Surfactant-like molecules reduce surface tension and prevent aggregation. This strategy is particularly useful in systems where some level of crystallization is unavoidable but deposition must be minimized.
In many production systems, a blended chemical package delivers the most reliable performance. Crystal modifiers reduce deposit strength, dispersants maintain suspension, and solvent components may assist in dissolving existing accumulations. The objective is not merely to prevent wax formation, which is thermodynamically inevitable below WAT, but to manage its behavior within the flow system.
Injection Strategy and Treatment Optimization
Chemical effectiveness depends heavily on proper placement and dosing. Injection points must be selected where mixing is sufficient to ensure uniform distribution before the fluid reaches critical cooling zones. In wellbores, this often means injecting upstream of temperature transition areas. In pipelines, treatment may be introduced at the wellhead or manifold to maximize residence time.
Dosage is determined by crude composition, wax content, flow rate, and temperature profile. Overdosing increases cost without proportional benefit, while underdosing leaves the system vulnerable. Laboratory testing, including cold finger tests and dynamic flow loop experiments, provides insight into optimal concentration levels.
Continuous injection programs typically provide better long-term control than batch treatments. However, in systems with intermittent flow or startup risks, batch injection may supplement routine dosing. Monitoring parameters such as differential pressure, pigging frequency, and production rate helps evaluate program effectiveness.
Importantly, chemical compatibility must be assessed with other treatment systems such as corrosion inhibitors, demulsifiers, or scale inhibitors. Poor compatibility can lead to emulsion stability changes or reduced overall performance. A holistic chemical management approach ensures flow assurance strategies operate in synergy rather than conflict.
Economic and Operational Implications
Wax control programs directly influence asset reliability. Unplanned shutdowns caused by wax blockages can cost operators substantial production losses per day. In offshore developments, intervention logistics amplify these costs significantly. Preventive chemical programs, though representing a recurring expense, often reduce total lifecycle cost when evaluated against downtime, mechanical cleaning, and equipment stress.
Energy efficiency also improves when pipelines remain free of restrictive deposits. Lower pressure drop translates into reduced pumping or compression demand. This not only lowers fuel consumption but also reduces greenhouse gas emissions associated with energy use.
From a risk management perspective, proactive wax inhibition enhances operational predictability. Production planning becomes more stable, maintenance schedules become more controlled, and asset lifespan extends. In increasingly competitive energy markets, such reliability provides measurable strategic advantage.
Wax Deposition in Subsea and Deepwater Systems
Subsea and deepwater developments present some of the most challenging environments for wax control. Unlike onshore systems where ambient temperatures may fluctuate seasonally, subsea pipelines are continuously exposed to near-freezing seawater temperatures. As hot reservoir fluids enter cold seabed tiebacks, temperature decline is rapid and sustained over long distances.
This extended cooling profile significantly increases the risk window for wax precipitation. In long subsea flowlines, the fluid may remain below Wax Appearance Temperature for hours before reaching processing facilities. During shutdown conditions, the risk intensifies further. Static cooling allows extensive crystallization, and restart operations can encounter partially solidified sections of pipeline.
Remediation in such environments is complex and costly. Mechanical pigging may not always be feasible, and thermal treatments require significant energy input. Chemical inhibition therefore becomes central to subsea flow assurance strategy. Continuous wax inhibitor injection at the wellhead or subsea manifold helps manage crystallization before fluids enter critical cold zones.
Designing subsea chemical programs requires detailed modeling of temperature profiles, flow regimes, and deposition rates. Computational flow assurance models simulate cooling curves and predict deposition thickness over time. These predictive tools allow engineers to optimize inhibitor dosage and injection frequency before field deployment.
Field Performance and Lessons from Mature Assets
Field data consistently demonstrates that early implementation of wax inhibition programs yields stronger long-term results than reactive cleanup strategies. Mature fields often experience shifting production profiles as reservoir pressure declines and water cut increases. These changes alter temperature gradients and fluid composition, sometimes intensifying wax deposition even in wells previously considered stable.
Operators who rely solely on periodic mechanical cleaning often observe recurring buildup shortly after intervention. This pattern reflects the underlying thermodynamic reality: if crude characteristics and temperature profiles remain unchanged, deposition will resume.
In contrast, proactive chemical programs that modify crystal growth dynamics tend to reduce deposit strength and adhesion. Even when some deposition occurs, it remains softer and easier to remove, reducing pigging frequency and minimizing pressure spikes.
Another lesson from field operations is the importance of adaptive dosing. Wax content and crude composition can vary over time. Regular laboratory testing of produced fluids ensures chemical programs remain aligned with evolving reservoir conditions. Static treatment strategies may underperform in dynamic production environments.
Integration with Monitoring and Digital Flow Assurance
Modern flow assurance increasingly incorporates digital monitoring tools. Temperature sensors, pressure monitoring systems, and real-time production analytics provide early warning of abnormal deposition patterns. A gradual increase in differential pressure along a pipeline often signals early wax accumulation.
When combined with predictive software, these measurements enable operators to anticipate deposition trends before blockages occur. Chemical injection rates can then be adjusted proactively rather than reactively.
Data-driven wax management reduces uncertainty. Instead of responding to emergencies, operators implement targeted adjustments based on measurable indicators. This integration of chemistry with digital surveillance reflects the broader industry shift toward intelligent asset management.
Emerging Trends in Wax Control Chemistry
As production systems become more complex, wax control chemistry continues to evolve. Advanced polymer technologies are being engineered to function across wider temperature ranges and higher shear conditions. These next-generation crystal modifiers offer improved adsorption efficiency and longer-lasting protection.
Research is also exploring nano-structured additives capable of interacting more precisely with paraffin molecules. Such materials aim to enhance dispersion and reduce the total chemical volume required for effective control.
Environmental considerations are driving formulation improvements as well. Modern inhibitor packages increasingly focus on biodegradability and reduced toxicity while maintaining performance under demanding field conditions. Sustainable chemistry development aligns wax management with evolving regulatory expectations.
In parallel, combined chemical strategies that address wax, corrosion, and scale simultaneously are gaining interest. Integrated treatment programs simplify logistics and reduce chemical compatibility issues, particularly in offshore developments where storage capacity is limited.

Conclusion
Wax deposition remains one of the most persistent organic flow assurance challenges in oil and gas production. Rooted in thermodynamic principles and influenced by temperature gradients, pressure changes, and crude composition, wax crystallization is an inevitable phenomenon once operating conditions cross critical thresholds.
However, inevitability does not equate to uncontrollability. By understanding the mechanisms of nucleation, crystal growth, aggregation, and surface adhesion, operators can design chemical strategies that manage deposition effectively. Wax inhibitors, pour point depressants, and dispersants modify crystallization behavior at the molecular level, reducing adhesion and maintaining flow stability.
In subsea and mature assets alike, proactive chemical programs consistently outperform reactive mechanical interventions. When integrated with monitoring systems and predictive modeling, wax management evolves from routine maintenance into strategic flow assurance planning.
As energy infrastructure extends into colder, deeper, and more complex environments, reliable wax control becomes increasingly essential. Chemical strategies that combine technical precision with operational foresight ensure uninterrupted production, lower lifecycle costs, and stronger asset integrity.
In modern oil and gas operations, wax control is no longer a seasonal concern or secondary maintenance issue. It is a core component of sustainable, efficient, and economically resilient production systems.
Frequently Asked Questions (FAQs)
1. What is wax deposition in oil and gas production?
Wax deposition refers to the accumulation of paraffin crystals on the inner walls of production tubing and pipelines when crude oil temperature drops below its Wax Appearance Temperature (WAT). These deposits restrict flow and increase pressure drop.
2. What causes wax to precipitate from crude oil?
Wax precipitates when temperature decreases below the crude’s solubility threshold for paraffin molecules. As fluids travel from hot reservoirs to cooler surface conditions, long-chain hydrocarbons crystallize and separate from the liquid phase.
3. What is Wax Appearance Temperature (WAT)?
Wax Appearance Temperature is the temperature at which wax crystals first begin to form in crude oil during cooling. It is a critical parameter for designing flow assurance and chemical treatment programs.
4. How does wax deposition affect production rates?
Wax buildup reduces pipeline diameter, increases frictional pressure losses, and forces artificial lift systems to work harder. In severe cases, it can cause partial or complete flow blockage.
5. Is wax deposition more common in subsea systems?
Yes. Subsea pipelines are exposed to cold seabed temperatures, accelerating crude cooling and increasing the duration fluids remain below WAT, which intensifies deposition risk.
6. How do wax inhibitors work?
Wax inhibitors modify crystal growth during nucleation. They prevent large, rigid crystals from forming and reduce adhesion to metal surfaces, allowing wax particles to remain dispersed in the fluid.
7. What is the difference between wax inhibitors and dispersants?
Wax inhibitors alter crystal structure to prevent adhesion, while dispersants keep precipitated wax particles suspended in the fluid to reduce aggregation and settlement.
8. Can mechanical pigging alone solve wax problems?
Pigging removes existing deposits but does not prevent reformation. Without chemical control, wax will continue to accumulate under the same thermodynamic conditions.
9. How is chemical dosage determined for wax control?
Dosage depends on crude composition, wax content, temperature profile, flow rate, and deposition severity. Laboratory testing such as cold finger and flow loop experiments help determine optimal concentration.
10. Why is proactive wax management more cost-effective?
Preventive chemical programs reduce shutdown frequency, lower intervention costs, decrease energy consumption, and extend asset life — ultimately reducing total lifecycle operational expenses.




