Introduction
Cementing operations in oil and gas wells represent one of the most critical stages of well construction. The primary objective of cement placement is to create a durable barrier that isolates geological formations, supports casing strings, and protects the well from fluid migration throughout its operational life. However, in modern drilling environments—especially in high-pressure, high-temperature (HPHT) wells—cement systems face significant operational challenges.
One of the most pressing issues in such environments is rapid cement setting. Elevated temperatures accelerate cement hydration reactions, reducing the time available for pumping, placement, and proper displacement of the slurry in the wellbore. If the cement sets prematurely before it reaches the intended zone, it can lead to incomplete zonal isolation, poor bonding, or even operational failures such as stuck casing or lost circulation.
To manage these challenges, engineers rely on specialized cement retarders—chemical additives designed to slow down the hydration process and extend the slurry’s workable time. Among the various retarding agents used in oilfield cementing, boric acid has emerged as a reliable and highly effective solution, particularly in high-temperature well conditions.
Boric acid plays a strategic role in controlling cement set times without compromising the mechanical strength or long-term durability of the cement sheath. By carefully regulating hydration reactions, it allows operators to maintain slurry pumpability, achieve accurate placement, and ensure proper cement bonding even under extreme downhole temperatures.
Understanding how boric acid functions within cement systems—and why it is especially valuable in HPHT wells—is essential for drilling engineers, cementing specialists, and production planners working in challenging reservoirs.
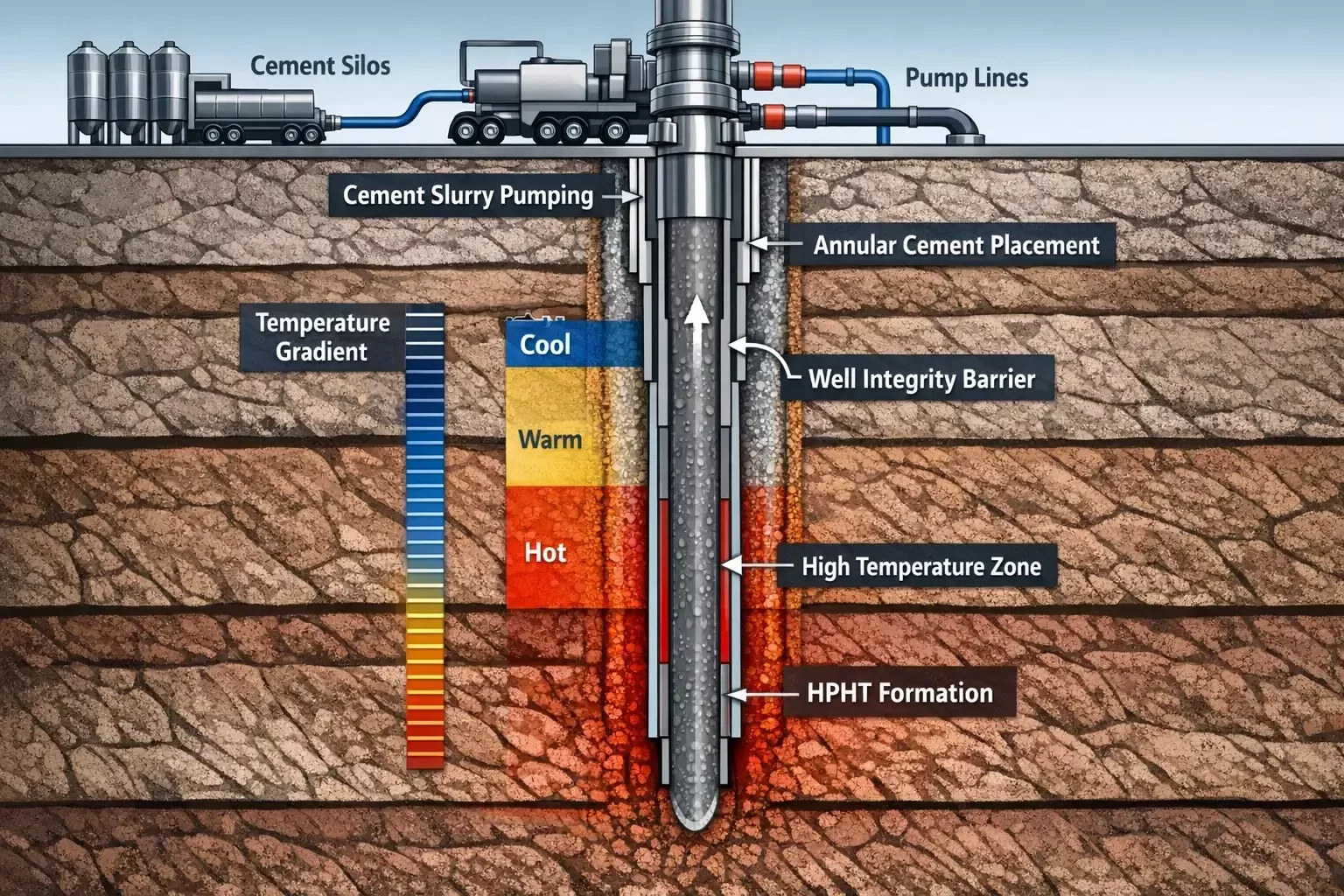
The Importance of Set Time Control in Oil Well Cementing
In oilfield cementing, the setting time of cement slurry is one of the most important parameters affecting operational success. The cement must remain fluid long enough to be pumped from the surface, displaced through the casing, and placed accurately in the annular space between the casing and the formation.
However, it must also be set within a predictable timeframe after placement to develop sufficient compressive strength and form an effective barrier.
Balancing these requirements becomes increasingly difficult as wells reach greater depths and encounter higher formation temperatures. In deep wells, bottomhole temperatures often exceed 120°C to 200°C, dramatically accelerating the chemical reactions responsible for cement hydration.
Under such conditions, standard cement formulations may begin to set far earlier than expected. This rapid thickening can reduce slurry pumpability and increase frictional pressure losses in the wellbore. If the slurry becomes too viscous during pumping operations, it may fail to reach the target zone, resulting in incomplete annular filling or poor displacement efficiency.
Poor cement placement can lead to several long-term operational risks, including gas migration, sustained casing pressure, or formation fluid communication between zones. These issues not only compromise well integrity but can also result in costly remedial cementing operations or production losses.
For these reasons, controlling cement set time is not simply a matter of operational convenience—it is a fundamental requirement for ensuring well safety, structural integrity, and long-term production performance.
Chemical retarders such as boric acid provide engineers with a reliable method to slow down hydration reactions and extend slurry pumpability without sacrificing final cement strength.
Challenges of Cementing in HPHT Wells
High-pressure, high-temperature wells present a unique set of technical challenges that significantly influence cementing design. As exploration moves deeper into complex reservoirs and offshore environments, these challenges are becoming increasingly common in modern drilling programs.
One of the defining characteristics of HPHT wells is the extreme thermal environment encountered during drilling and completion operations. Temperatures exceeding 150°C can accelerate cement hydration reactions to such an extent that conventional cement systems may begin setting within minutes rather than hours.
This rapid setting behavior limits the available time for slurry placement and increases the risk of operational complications during pumping.
Another major challenge involves pressure stability and formation integrity. HPHT reservoirs often require higher-density cement slurries to maintain hydrostatic pressure control and prevent formation fluid influx. However, heavier slurries typically exhibit higher viscosity and shorter pumpability windows, further complicating placement operations.
Additionally, the long casing strings associated with deep wells increase the time required to pump cement from the surface to the target zone. If the slurry begins to thicken prematurely, the pressure required to continue pumping may exceed safe operating limits, potentially damaging the formation or causing equipment stress.
Temperature gradients within the wellbore also create variations in cement hydration rates. While the slurry may remain stable at surface conditions, it can react far more rapidly once exposed to elevated bottomhole temperatures.
These combined factors make precise control of cement hydration kinetics essential in HPHT cementing programs. Retarding agents such as boric acid help address this challenge by slowing down the chemical reactions responsible for cement setting, thereby ensuring adequate pumping time and reliable placement.
Understanding Cement Hydration in Oilfield Applications
To appreciate the role of boric acid in cement systems, it is important to understand the basic chemistry of cement hydration.
Oil well cement is typically composed of Portland cement clinker, which contains several mineral compounds including tricalcium silicate, dicalcium silicate, tricalcium aluminate, and tetracalcium aluminoferrite. When water is added to the cement powder, these compounds undergo hydration reactions that gradually transform the slurry into a hardened solid matrix.
During the early stages of hydration, the slurry remains fluid and pumpable. As chemical reactions progress, the formation of calcium silicate hydrate (C-S-H) and other compounds causes the mixture to thicken and eventually set into a rigid structure.
Temperature plays a critical role in this process. Higher temperatures accelerate the rate of chemical reactions, causing the cement to set faster. While this may be beneficial in shallow wells where rapid strength development is desirable, it can be problematic in deep HPHT wells where longer pumpability windows are required.
Without proper chemical control, accelerated hydration can cause the slurry to lose its flow properties before it reaches the intended placement zone.
Boric acid functions as a cement retarder by interfering with these hydration reactions, particularly those involving calcium ions and aluminates. By slowing down the formation of early hydration products, boric acid delays the thickening process and extends the working time of the cement slurry.
This controlled retardation allows engineers to maintain stable slurry rheology during pumping while still achieving adequate strength development once the cement has been placed.
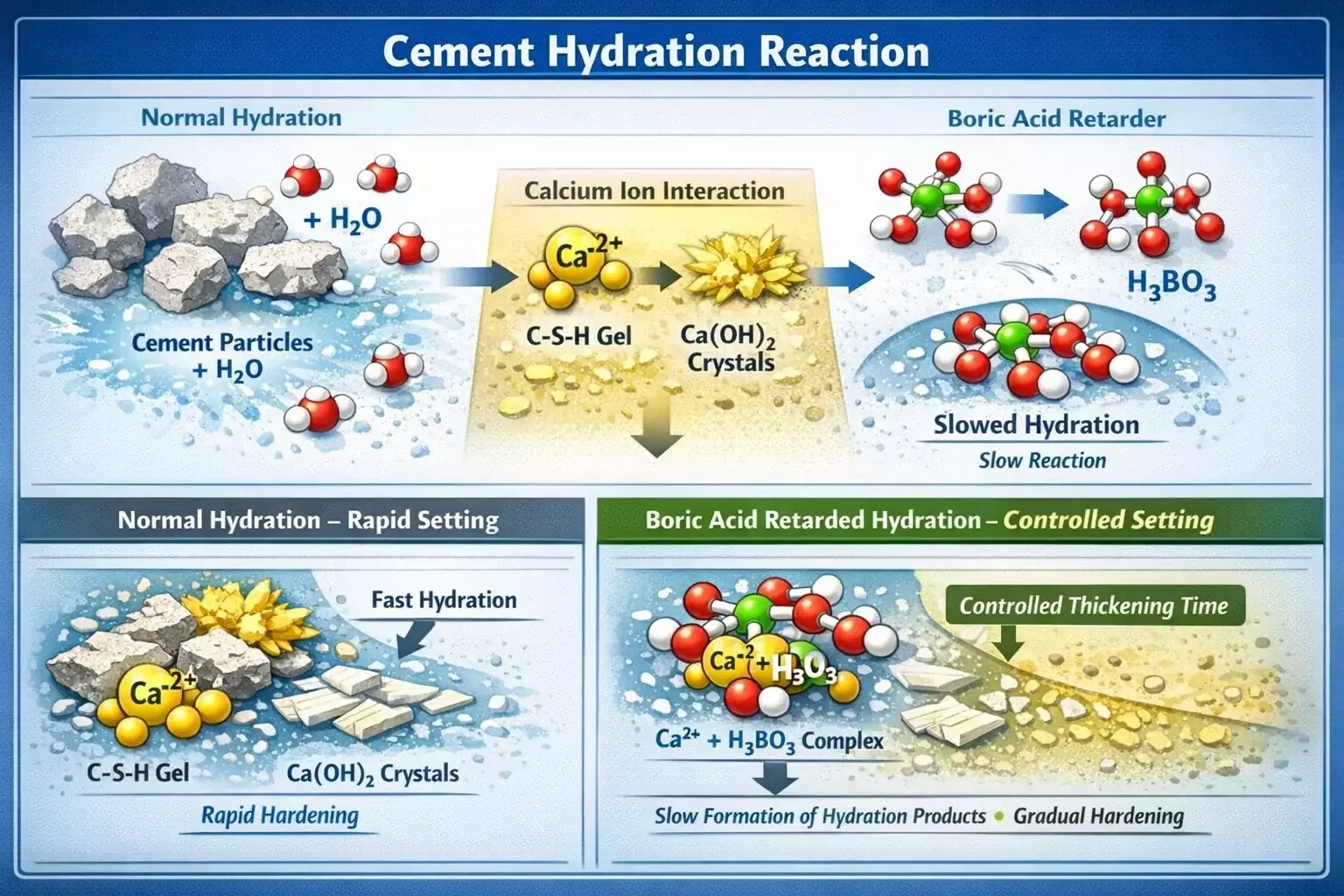
Chemical Properties of Boric Acid in Cement Systems
Boric acid (H₃BO₃) is a weak inorganic acid commonly used in industrial chemistry due to its buffering properties and ability to influence ionic reactions. In oilfield cementing systems, it functions primarily as a set retarder, meaning it delays the rate at which cement hydration reactions occur.
Unlike some organic retarders that rely on complex molecular interactions, boric acid works through relatively straightforward chemical mechanisms involving calcium ion interactions and the stabilization of hydration products. These interactions allow the cement slurry to maintain its pumpable state for a longer duration, even under elevated temperature conditions.
In cement slurry formulations, boric acid dissolves in the aqueous phase and begins interacting with the cement’s mineral compounds, particularly those associated with early hydration reactions. The compound forms complexes with calcium ions present in the slurry, effectively slowing down the formation of hydration products responsible for cement thickening.
This controlled interference with hydration chemistry allows engineers to extend slurry thickening time without significantly altering the overall cement composition. Because boric acid is effective at relatively low concentrations, it can be integrated into cement systems without dramatically changing slurry rheology or density.
Another important characteristic of boric acid is its thermal stability. Many organic retarders lose effectiveness or degrade when exposed to very high temperatures, especially in HPHT wells. Boric acid, however, remains chemically stable across a wide temperature range, making it particularly valuable for deep-well cementing operations where bottomhole temperatures may exceed 150°C.
Its predictable chemical behavior and compatibility with common cement additives have made boric acid a reliable component in cement formulations designed for challenging drilling environments.
Mechanism of Hydration Retardation
The effectiveness of boric acid as a cement retarder lies in its ability to control the early-stage reactions responsible for cement thickening.
During cement hydration, calcium ions released from cement minerals react with water to form various compounds that gradually transform the slurry into a hardened matrix. Among these reactions, those involving tricalcium aluminate are particularly rapid and contribute significantly to early setting behavior.
Boric acid interacts with these calcium ions to form borate complexes, which slow down their participation in hydration reactions. This interaction effectively delays the formation of calcium silicate hydrate and other compounds responsible for early cement stiffening.
Another important aspect of boric acid’s action is its ability to form a temporary protective layer around cement particles. This layer slows the penetration of water into the cement grains, thereby reducing the rate at which hydration reactions occur.
By controlling these processes, boric acid extends the time during which the slurry remains fluid and pumpable. The delay allows the cement to be transported safely through long casing strings and positioned accurately in the annulus before the setting process begins.
Importantly, the retardation effect is temporary and controlled. Once the slurry has been placed and the retarding influence gradually diminishes, hydration reactions resume and the cement develops the compressive strength necessary for structural support and zonal isolation.
This balance between delayed hydration and eventual strength development is critical in ensuring that cement systems perform reliably in high-temperature wells.
Compatibility with Other Cement Additives
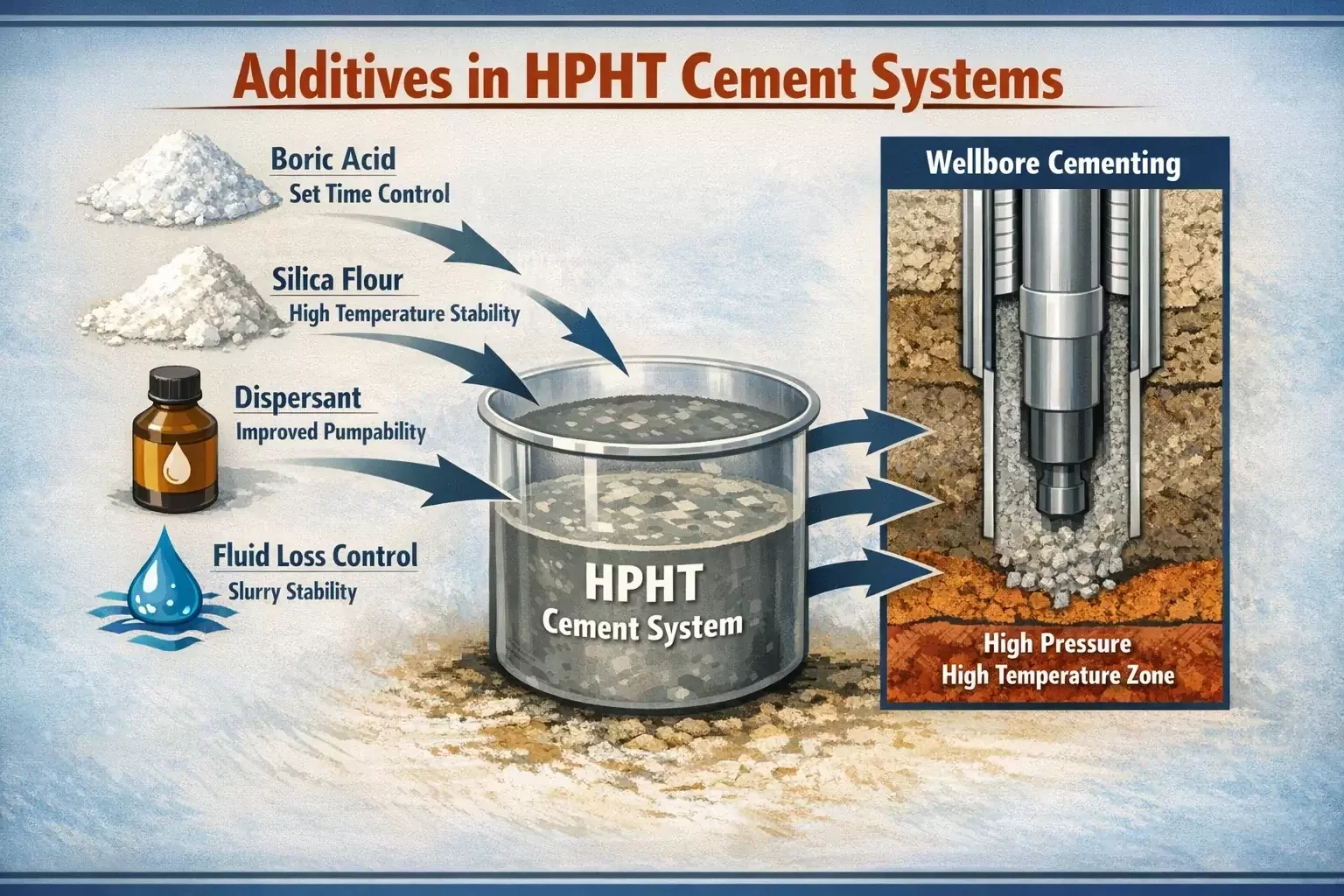
Modern oil well cement systems rarely rely on a single additive. Instead, they are carefully engineered blends containing multiple chemical components designed to address specific operational challenges. Boric acid is particularly valuable because it exhibits strong compatibility with many of these additives, allowing it to function effectively within complex cement formulations.
In HPHT wells, silica flour is frequently added to cement slurries to prevent strength retrogression at elevated temperatures. Silica flour reacts with calcium hydroxide to form thermally stable hydration products that maintain compressive strength under high-temperature conditions. Boric acid works well alongside silica flour, allowing both additives to perform their respective roles without interfering with each other’s functionality.
Dispersants are another common additive used to improve slurry flow properties and reduce viscosity. By dispersing cement particles more evenly throughout the slurry, these agents help maintain pumpability and minimize friction during cement placement. Boric acid can be incorporated into dispersant-containing systems without significantly affecting their rheological performance.
Fluid loss control agents are also frequently included in cement formulations to prevent excessive water loss into porous formations. Excessive fluid loss can lead to premature thickening and incomplete cement placement. Boric acid complements these additives by ensuring that hydration reactions remain slow enough to maintain slurry stability throughout the pumping process.
Because of this compatibility with other additives, boric acid can be integrated into highly customized cement systems tailored to specific well conditions. Engineers can adjust additive concentrations to achieve precise control over thickening time, slurry density, and mechanical strength development.
This flexibility is particularly valuable in HPHT wells where multiple operational variables—such as temperature gradients, pressure conditions, and formation characteristics—must be carefully managed to achieve successful cement placement.
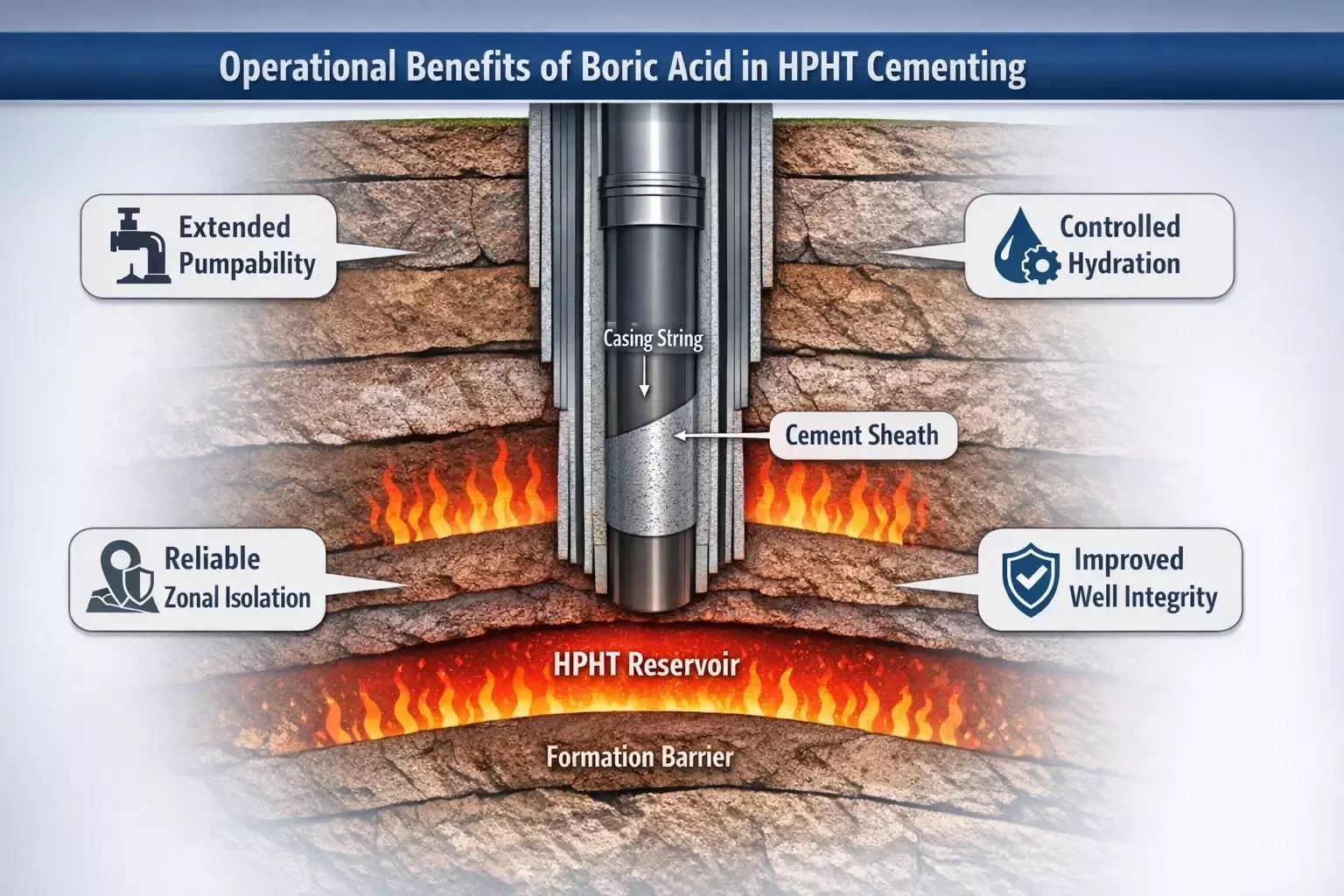
Operational Benefits in High-Temperature Cementing
The use of boric acid in cement systems provides several operational advantages that directly contribute to the success of cementing operations in challenging environments.
One of the most significant benefits is the ability to extend thickening time without excessively increasing slurry viscosity. Maintaining manageable viscosity levels is crucial for ensuring that cement slurries can be pumped efficiently through long wellbores and complex casing geometries.
Extended pumpability also improves displacement efficiency during cement placement. When the slurry remains fluid for a longer period, it can more effectively displace drilling fluids and fill irregularities in the annular space. This improves the likelihood of achieving a continuous cement sheath capable of isolating formations and supporting casing strings.
Another advantage involves operational safety and reliability. Premature cement setting can lead to equipment damage, stuck pipe incidents, or incomplete cement placement. By controlling hydration reactions, boric acid reduces the likelihood of these complications, helping maintain smooth and predictable cementing operations.
In deep and offshore wells, where logistical costs and operational risks are significantly higher, the ability to maintain stable cement slurry behavior becomes even more critical. Boric acid provides engineers with a reliable tool for managing these challenges while preserving the structural integrity of the final cement sheath.
Boric Acid Performance in HPHT Field Environments
In high-pressure, high-temperature wells, cementing operations must be designed with exceptional precision. The deeper the well, the greater the thermal stress imposed on cement systems. Bottomhole temperatures in HPHT reservoirs frequently exceed 150°C and in some cases approach 200°C, significantly accelerating hydration reactions within conventional cement slurries.
Under these conditions, cement that might normally remain pumpable for several hours at surface temperature can begin to thicken rapidly once it enters the high-temperature sections of the wellbore. Without the use of effective retarders, the slurry can lose mobility before it reaches the intended annular zone.
Boric acid helps mitigate this risk by delaying the hydration reactions responsible for cement thickening, allowing sufficient time for the slurry to be pumped through long casing strings and placed accurately around the casing.
In offshore wells and deep onshore drilling projects, cement often must travel thousands of meters before reaching the target placement zone. The temperature gradient encountered during this journey means the slurry experiences increasingly aggressive thermal conditions as it moves downward. Boric acid helps maintain a stable hydration rate throughout this temperature transition.
Field experience has shown that cement systems containing boric acid demonstrate predictable thickening time behavior even under elevated temperatures, enabling operators to plan pumping schedules with greater accuracy.
This predictability is crucial for ensuring proper displacement of drilling fluids and achieving complete annular coverage. A well-placed cement sheath forms the structural backbone of well integrity, preventing unwanted fluid movement between geological formations.
Cement Design Considerations in HPHT Wells
Designing cement systems for HPHT wells requires a careful balance of several competing factors. Engineers must consider temperature effects, hydrostatic pressure requirements, slurry density, rheology, and long-term mechanical strength.
Boric acid is incorporated into cement formulations as part of a broader additive package designed to address these complex challenges.
One of the most important considerations is thickening time control. Cement slurries must remain pumpable long enough to allow for placement, but they must also develop compressive strength within a reasonable timeframe after placement to allow drilling operations to proceed.
Boric acid helps regulate the early hydration reactions that determine this thickening window. By delaying the initial chemical reactions within the slurry, it extends the operational pumping period without preventing the cement from ultimately setting and developing strength.
Another design factor involves temperature gradients along the wellbore. Cement slurries often encounter dramatically different temperatures at the surface compared to bottomhole conditions. A retarder such as boric acid must therefore perform reliably across a broad temperature range.
HPHT cement systems frequently incorporate additional additives alongside boric acid. For example, silica flour is often included to prevent strength retrogression in high-temperature environments. Dispersants may be added to control slurry viscosity and improve flow characteristics during pumping.
Fluid loss control agents help maintain slurry stability by preventing excessive water loss into surrounding formations. Together, these additives create a cement system capable of performing reliably in extreme downhole conditions.
Boric acid’s compatibility with these additives allows engineers to design cement systems that maintain both extended pumpability and long-term mechanical integrity.
Impact on Zonal Isolation and Well Integrity
The ultimate goal of oil well cementing is to establish effective zonal isolation. The cement sheath placed between the casing and the formation acts as a permanent barrier that prevents the movement of fluids between different geological layers.
If cement placement is incomplete or poorly bonded, fluids such as gas, oil, or water may migrate through the annular space, potentially causing serious operational problems. These issues can include sustained casing pressure, cross-flow between formations, or environmental contamination.
Premature cement setting is one of the primary causes of poor cement placement. If the slurry begins to thicken before it has fully displaced drilling fluids or filled the annular space, voids or channels may remain in the cement sheath.
Boric acid helps prevent this scenario by extending the slurry’s pumpability window, allowing it to flow smoothly through the wellbore and fill irregularities in the annulus.
Proper placement ensures that the cement forms a continuous and uniform barrier capable of supporting casing strings and maintaining zonal isolation over the entire life of the well.
In HPHT wells, where extreme temperature and pressure conditions place additional stress on the cement sheath, maintaining strong bonding and structural integrity becomes even more critical.
The controlled retardation provided by boric acid allows cement systems to develop strength gradually and uniformly, reducing the risk of micro-annulus formation or structural weaknesses within the cement matrix.
This contributes directly to long-term well stability, production safety, and environmental protection.
Operational Reliability in Deep and Offshore Wells
Deepwater and offshore drilling environments present additional logistical and operational challenges for cementing operations. The extended length of casing strings, combined with complex well geometries, increases the time required to pump cement from the surface to the target depth.
In such scenarios, the margin for error becomes extremely small. If the cement slurry begins to thicken prematurely, the entire operation may be compromised.
Boric acid provides a valuable layer of operational security by ensuring that cement systems maintain their fluidity during the critical pumping phase.
The ability to extend thickening time allows operators to perform necessary operational steps—such as displacement, circulation, and pressure monitoring—without the risk of the slurry setting too early.
Furthermore, deep offshore wells often involve significant financial investment. Any failure in cementing operations may require costly remedial work, including squeeze cementing or even sidetracking the well.
By improving slurry stability and placement accuracy, boric acid helps reduce the likelihood of such complications and supports more reliable cementing outcomes in technically demanding drilling environments.
Operational Advantages of Boric Acid in Modern Cementing Programs
In modern drilling operations, cementing programs must be carefully designed to ensure both operational efficiency and long-term well integrity. As wells become deeper and reservoirs more technically complex, cement additives play an increasingly strategic role in maintaining reliable cement placement and performance.
Boric acid offers several operational advantages that make it particularly valuable in challenging cementing environments.
One of its primary benefits is predictable thickening time control. Unlike some organic retarders that can behave unpredictably at extreme temperatures, boric acid provides consistent retardation performance across a wide range of thermal conditions. This predictability allows engineers to design cement systems with greater confidence, ensuring that pumping schedules remain stable and operational risks are minimized.
Another advantage lies in its chemical stability under HPHT conditions. High temperatures can degrade certain additives, reducing their effectiveness during cementing operations. Boric acid maintains its retarding properties even in elevated thermal environments, making it a reliable component in deep and offshore well cement formulations.
Boric acid also contributes to improved slurry stability. By controlling the early hydration reactions within the cement, it prevents premature thickening and allows the slurry to maintain a uniform rheological profile throughout the pumping process. This helps ensure that the cement flows evenly through the wellbore and fills the annular space effectively.
From an operational perspective, these properties reduce the likelihood of complications such as stuck casing, incomplete displacement, or early cement setting—issues that can significantly disrupt drilling schedules and increase operational costs.
Comparison with Other Cement Retarders
Various chemical retarders are used in oilfield cementing systems, including lignosulfonates, organic acids, and synthetic polymer-based additives. Each of these retarders has specific advantages depending on the well environment and cement design requirements.
However, in high-temperature wells, certain organic retarders may lose effectiveness due to thermal degradation. As temperature increases, their chemical structures may break down, reducing their ability to control hydration reactions.
Boric acid offers a distinct advantage in this regard because of its thermal resilience and stable chemical behavior. Its retarding mechanism relies primarily on interactions with calcium ions and cement hydration products, rather than complex organic molecular structures that may degrade at high temperatures.
Another advantage of boric acid is its compatibility with silica-rich cement systems commonly used in HPHT wells. Since silica flour is frequently added to prevent strength retrogression at elevated temperatures, the retarder used must function effectively within this environment.
Boric acid works well alongside silica-based additives and does not significantly interfere with the development of long-term compressive strength.
Additionally, boric acid can be used in relatively small concentrations, making it efficient from both chemical and operational perspectives. Engineers can achieve significant retardation effects without dramatically altering slurry density or viscosity.
These characteristics make boric acid a practical and dependable option in many high-temperature cementing programs.
The Role of Chemical Engineering in Advanced Cement Systems
The continued development of advanced cement additives reflects the growing importance of chemical engineering in modern oil and gas operations.
As exploration expands into deeper and more complex reservoirs, well conditions are becoming increasingly demanding. High temperatures, extreme pressures, and extended wellbore lengths require cement systems that are capable of maintaining both pumpability and long-term durability.
Chemical additives such as boric acid allow engineers to fine-tune cement hydration reactions and adapt cement systems to specific operational conditions. This level of control helps ensure that cementing operations remain reliable even in challenging environments.
Furthermore, advances in cement chemistry are enabling the development of customized additive packages tailored to specific well conditions. Retarders, dispersants, fluid loss agents, and strength enhancers can be combined in precise proportions to create cement systems capable of meeting the unique demands of each drilling project.
Within this framework, boric acid continues to serve as an important component in cement formulations designed for HPHT wells.
Conclusion
Cementing operations form the structural foundation of oil and gas wells, providing the barriers necessary to isolate formations, support casing strings, and maintain long-term well integrity. In high-pressure, high-temperature wells, controlling cement hydration reactions becomes particularly critical due to the accelerated setting behavior caused by elevated temperatures.
Boric acid plays a strategic role in addressing this challenge by functioning as an effective cement retarder. Through its interactions with calcium ions and hydration reactions, it slows the rate at which cement thickens, allowing the slurry to remain pumpable for a longer period during placement operations.
This controlled retardation helps ensure accurate cement placement, improved displacement efficiency, and the formation of a continuous cement sheath capable of providing reliable zonal isolation.
Its chemical stability, compatibility with other additives, and predictable performance in high-temperature environments make boric acid a valuable tool in modern cementing programs.
As drilling operations continue to push into deeper and more technically complex reservoirs, the importance of advanced cement chemistry will only continue to grow. Retarding agents such as boric acid will remain essential in helping engineers manage hydration kinetics, maintain operational efficiency, and ensure the long-term integrity of wells operating under extreme conditions.




