
Introduction: The Invisible Role of Dehydration in Gas Operations
Natural gas rarely leaves the reservoir in a clean, dry state. Along with hydrocarbons, it carries water vapor, condensed liquids, and dissolved contaminants. If that moisture is not removed before transport, it can cause hydrate formation, corrosion, pipeline blockages, and severe operational disruptions. Gas dehydration is therefore not a downstream convenience — it is a core requirement for safe production and transmission.
In modern gas processing, glycols are the backbone of dehydration systems. Among them, monoethylene glycol (MEG) and triethylene glycol (TEG) are the two most widely used. At first glance, they appear similar. Both are clear liquids, both absorb water, and both circulate through regeneration loops. Yet in practice, they serve very different roles, operate under different philosophies, and solve different engineering problems.
Choosing between MEG and TEG is not a simple substitution decision. It is a strategic selection that affects hydrate control, energy consumption, regeneration complexity, logistics, corrosion behavior, and long-term operating cost. Understanding their differences is essential for engineers designing dehydration systems in upstream, midstream, and offshore facilities.
This blog examines MEG and TEG not as competing chemicals, but as specialized tools. Each has a defined place in gas processing, and selecting the correct one depends on understanding how dehydration objectives align with system design.
Why Water Removal Is Critical in Gas Systems
Water in natural gas is more than an impurity; it is an operational hazard. When pressure and temperature conditions shift during transport, water can combine with hydrocarbons to form gas hydrates — crystalline solids that behave like ice but form at much higher temperatures. Hydrates can block pipelines, choke valves, damage compressors, and trigger emergency shutdowns.
Beyond hydrate formation, water accelerates corrosion inside pipelines and processing equipment. In the presence of carbon dioxide or hydrogen sulfide, condensed water becomes acidic and aggressively attacks carbon steel. This creates internal corrosion risks that compromise asset integrity.
Moisture also interferes with downstream processing. Liquefaction, fractionation, and petrochemical conversion all require tightly controlled gas specifications. Even small amounts of water vapor can reduce efficiency or contaminate end products.
Because of these risks, dehydration is not optional. It is a protective barrier that ensures flow assurance, equipment longevity, and compliance with pipeline specifications.
The Role of Glycols in Dehydration Systems
Glycol dehydration systems operate on a simple but powerful principle: selective absorption. Glycols have a strong affinity for water molecules. When wet gas contacts glycol in an absorber column, water transfers from the gas phase into the liquid glycol. The dried gas exits the top of the tower, while the water-rich glycol is regenerated and recycled.
This cycle repeats continuously, allowing large volumes of gas to be processed with relatively compact equipment. The success of this system depends heavily on the properties of the glycol used. Boiling point, vapor pressure, viscosity, thermal stability, and regeneration behavior all influence how effectively the system performs.
MEG and TEG both absorb water efficiently, but they are optimized for different operating conditions. One is designed for hydrate inhibition in flowlines and subsea systems. The other is designed for deep dehydration in processing plants. Understanding this distinction is the key to choosing correctly.
What Is MEG? A Hydrate Control Specialist
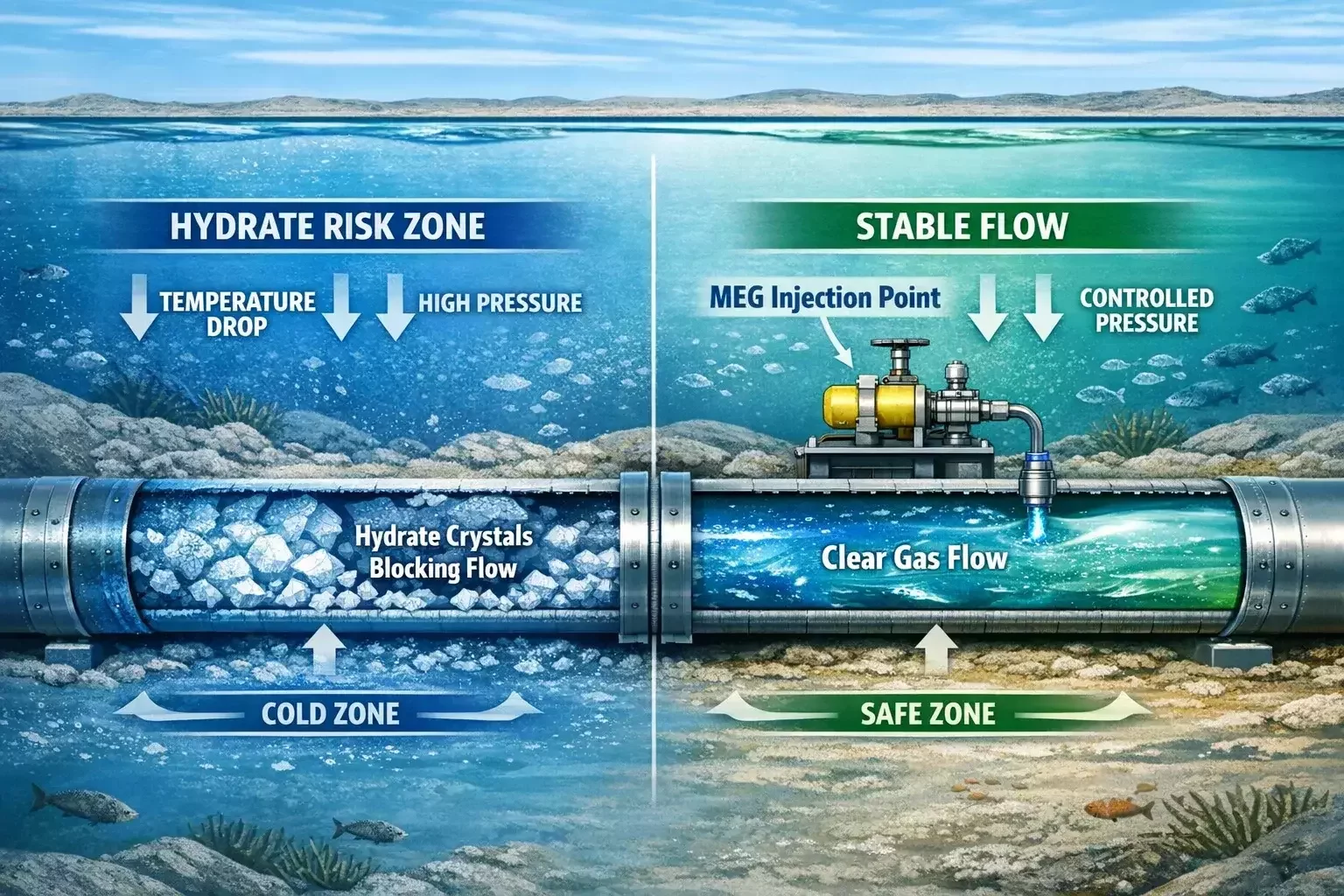
Monoethylene glycol is primarily associated with hydrate prevention rather than conventional dehydration. In many gas production systems — especially offshore and subsea fields — MEG is injected directly into pipelines to suppress hydrate formation before it can occur.
Instead of removing water from the gas completely, MEG changes the thermodynamic conditions so hydrates cannot form. It acts as an antifreeze agent inside the pipeline. After flowing with the produced fluids, the MEG-water mixture is recovered at processing facilities, regenerated, and reinjected.
This makes MEG systems part of a closed-loop chemical management strategy. They are designed not just for dehydration performance, but for chemical recovery efficiency, contamination tolerance, and large-scale circulation.
MEG is especially valuable in long subsea tiebacks where traditional dehydration equipment cannot be placed near the wellhead. In such environments, hydrate prevention is more practical than full dehydration at the source.
What Is TEG? The Industry Standard for Gas Drying
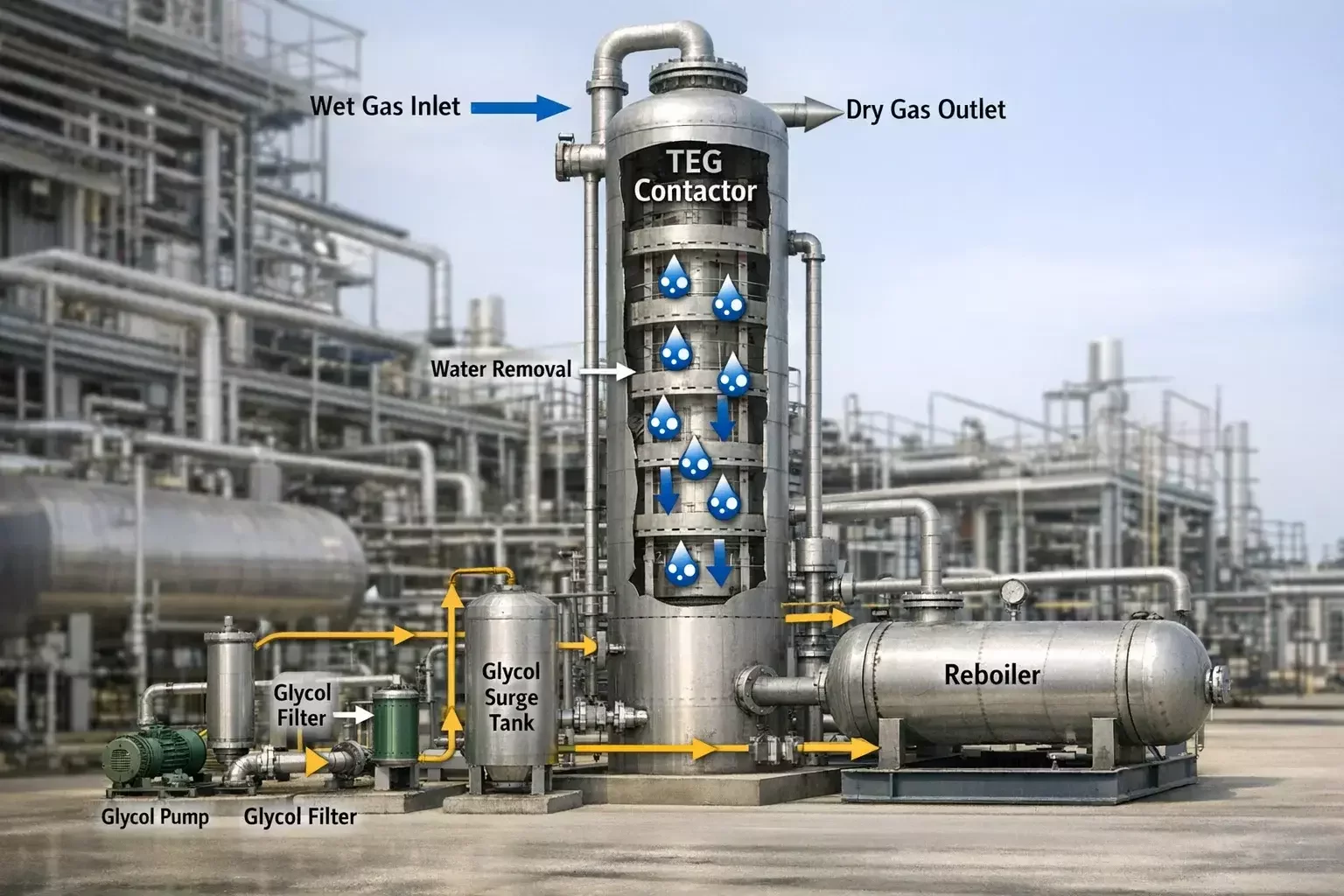
Triethylene glycol serves a different mission. TEG systems are designed to deeply dry gas to meet pipeline and processing specifications. Unlike MEG, which modifies hydrate conditions, TEG physically removes water vapor from the gas stream.
TEG dehydration units are common in gas plants, gathering stations, and transmission hubs. These systems achieve very low water content, producing gas suitable for long-distance transport and downstream processing.
TEG is favored because of its high boiling point and low vapor pressure, which allow efficient regeneration without excessive glycol loss. It can be heated to remove absorbed water while remaining stable enough for continuous reuse.
Where MEG is a hydrate control fluid circulating through production flowlines, TEG is a dehydration workhorse operating inside fixed processing equipment. Each chemical is optimized for its specific role.
Why Engineers Must Understand the Difference
Confusing MEG and TEG leads to design inefficiencies. A system built for deep dehydration will not perform well if treated as a hydrate inhibitor, and a hydrate loop will fail if designed like a traditional TEG plant.
The decision is not about which glycol is “better.” It is about matching chemical behavior to operational objectives. Gas composition, transport distance, pressure, temperature, infrastructure layout, and recovery logistics all influence the correct choice.
In the next section, we will examine the technical performance differences between MEG and TEG in detail — including thermodynamics, regeneration energy, corrosion behavior, and operational trade-offs. This deeper comparison reveals why each glycol dominates specific segments of the gas industry.
Absorption Performance: How MEG and TEG Handle Water Differently
At the heart of glycol dehydration is water absorption efficiency, but MEG and TEG approach this task from different thermodynamic directions. TEG is engineered for deep dehydration. Its molecular structure allows it to absorb large quantities of water while maintaining low vapor losses, making it ideal for achieving pipeline-grade dryness.
TEG systems are designed around equilibrium absorption. Wet gas enters an absorber tower and contacts lean TEG in a counter-current flow. The glycol pulls water vapor from the gas until equilibrium is reached. Because TEG has a high affinity for water and a low vapor pressure, it can remove moisture to extremely low levels when regenerated properly.
MEG behaves differently. It absorbs water effectively, but its role is not to dry gas to specification. Instead, it lowers the freezing point of the water phase in multiphase flow. MEG systems tolerate higher water content because their objective is hydrate suppression, not ultra-dry gas production. This distinction shapes everything about system design, from circulation rates to regeneration targets.
In practical terms, TEG systems chase dryness, while MEG systems chase stability.
Regeneration Requirements: Energy and Infrastructure Differences
The most significant engineering difference between MEG and TEG systems lies in regeneration. TEG dehydration units operate at high temperatures to boil off absorbed water and restore glycol purity. Standard TEG reboilers run near the thermal stability limit of the glycol, often exceeding 200°C. This high-temperature regeneration is energy intensive but necessary to achieve lean glycol concentrations suitable for deep dehydration.
MEG regeneration operates under a different philosophy. Because MEG systems are part of a closed-loop hydrate control program, regeneration must handle contaminants in addition to water. Produced fluids introduce salts, hydrocarbons, organic acids, and solids into the MEG loop. Over time, these impurities accumulate and reduce efficiency.
As a result, MEG regeneration plants are more complex. They often include flash separation, filtration, salt removal units, and reclaiming systems to maintain chemical purity. While TEG units focus on thermal efficiency, MEG facilities emphasize contaminant management and chemical recovery.
This difference has major implications for capital investment. TEG plants are typically smaller and more compact. MEG regeneration systems resemble miniature processing plants, especially in offshore developments where chemical recovery is critical for cost and environmental reasons.
Circulation Rates and System Scale
Another important distinction is circulation philosophy. TEG systems operate with relatively low circulation rates because their purpose is targeted dehydration within absorber columns. The glycol remains inside the plant and is continuously regenerated on-site.
MEG systems circulate much larger volumes of chemical. Because MEG is injected directly into flowlines, it travels long distances with produced fluids before being recovered. Circulation rates must account for transport losses, dilution, and recovery efficiency. This makes MEG systems more sensitive to logistics, storage capacity, and chemical inventory management.
In offshore fields, the economics of MEG depend heavily on recovery percentage. Even small losses translate into significant chemical costs over time. Engineers must therefore design recovery and regeneration systems that maximize reuse while minimizing waste.
Thermal Stability and Degradation Behavior
Thermal stability is another critical factor. TEG is stable at high regeneration temperatures, but prolonged exposure to oxygen or excessive heat can cause degradation. Degraded TEG forms acids and byproducts that increase corrosion risk and reduce absorption efficiency. Proper oxygen control and temperature management are essential to maintain glycol life.
MEG, while regenerated at lower temperatures, faces a different challenge: contamination rather than thermal breakdown. Salts and organic compounds entering the MEG loop can cause fouling, scaling, and foaming inside regeneration equipment. Without proper treatment, these impurities reduce heat transfer efficiency and damage system reliability.
This is why MEG programs require robust chemical housekeeping. Filtration, reclaiming, and periodic system cleaning are not optional — they are central to maintaining performance.
Corrosion Implications of Each System
Both glycols influence corrosion behavior, but in different ways. TEG dehydration units operate in controlled plant environments where oxygen ingress can be minimized. When properly maintained, TEG systems present relatively low corrosion risk. However, degradation products from overheated glycol can become corrosive if not monitored.
MEG systems operate in more chemically aggressive environments. Because MEG travels through production flowlines, it encounters CO₂, H₂S, salts, and microorganisms. This creates conditions where corrosion control becomes intertwined with glycol management.
Operators often combine MEG programs with corrosion inhibitors and biocides to protect infrastructure. The glycol itself is not inherently corrosive, but the environment it travels through can be.
Operational Flexibility and Field Suitability
TEG dehydration units are best suited for fixed installations where gas processing occurs in centralized facilities. They are efficient, predictable, and widely understood. Their footprint and energy requirements make them ideal for onshore plants and large offshore platforms with established infrastructure.
MEG systems excel in remote or subsea developments where dehydration at the wellhead is impractical. Long tiebacks, deepwater production, and cold environments favor MEG because hydrate prevention is more reliable than transporting wet gas untreated.
In essence, TEG belongs to processing plants, while MEG belongs to flow assurance systems.
Economic Trade-Offs
From a cost perspective, the decision is nuanced. TEG units require high regeneration energy but lower chemical inventory. MEG systems require large chemical volumes and complex recovery infrastructure but reduce hydrate risk over long distances.
Capital expenditure, operating expenditure, chemical loss tolerance, and field layout all influence which glycol becomes economically favorable. There is no universal winner — only context-driven optimization.
Offshore Developments: Why MEG Dominates Long Tiebacks
Offshore gas production presents some of the harshest operating conditions in the energy industry. Subsea pipelines are exposed to low seabed temperatures and high pressures — exactly the environment where hydrates thrive. Once hydrates form, they can completely block flowlines, requiring costly shutdowns or intervention campaigns that are both dangerous and expensive.
In long-distance subsea tiebacks, dehydration at the wellhead is rarely practical. Equipment footprint, maintenance access, and safety constraints make large TEG units difficult to deploy near the reservoir. This is where MEG becomes the preferred strategy.
MEG is injected directly into the multiphase stream at the subsea tree or wellhead. Instead of trying to remove water, the system chemically prevents hydrate formation during transport. The produced fluids carry MEG back to the host facility, where it is recovered, cleaned, and reinjected.
This closed-loop approach allows operators to transport wet gas over tens or even hundreds of kilometers without hydrate blockage. For deepwater and ultra-deepwater fields, MEG is often the only economically viable flow assurance solution.
Onshore Processing Plants: Where TEG Excels
Onshore facilities operate under a different set of constraints. Here, gas is typically processed at centralized plants with access to utilities, maintenance crews, and energy supply. In this environment, TEG dehydration becomes highly efficient.
TEG systems are compact relative to their dehydration capacity and can consistently deliver gas that meets strict pipeline moisture specifications. Because infrastructure is stable and regeneration energy is available, operators can optimize TEG units for long-term reliability.
Onshore plants also benefit from easier monitoring and maintenance. Operators can adjust glycol purity, temperature control, and inhibitor programs in real time, ensuring performance stability over decades of operation.
In many cases, onshore systems use a hybrid strategy: MEG is employed upstream for flow assurance, and TEG completes the dehydration process at the processing plant. This layered approach demonstrates how the two glycols are not competitors, but complementary tools.
Subsea Compression and Emerging Architectures
Modern field development is pushing technology into new territory. Subsea compression systems, floating production units, and extended step-out developments demand innovative dehydration strategies.
In these architectures, engineers must balance footprint, energy consumption, chemical logistics, and recovery efficiency. MEG recovery plants are becoming more advanced, incorporating salt removal, reclaiming technology, and energy integration to reduce operating costs.
Meanwhile, next-generation TEG systems are being optimized with heat recovery, advanced stripping gas systems, and digital monitoring to improve dehydration efficiency while lowering fuel consumption.
The industry trend is clear: glycol systems are becoming smarter, cleaner, and more integrated with digital asset management.
Decision Framework: How Engineers Choose Between MEG and TEG
Selecting the correct glycol is not a simple technical preference — it is a systems-level decision influenced by geology, infrastructure, economics, and safety.
Engineers begin by asking fundamental questions:
Is the primary risk hydrate formation or water content in export gas?
How long is the transport distance before processing?
What is the operating temperature and pressure envelope?
Is chemical recovery feasible?
What infrastructure exists for regeneration?
If hydrate risk dominates and dehydration cannot occur immediately, MEG becomes the logical choice. If the goal is meeting strict gas dryness specifications in a centralized facility, TEG is typically superior.
Many developments use both. MEG handles flow assurance upstream, and TEG completes dehydration downstream. The key is understanding where each glycol delivers maximum value.
Environmental and Sustainability Considerations
Modern oil and gas projects are increasingly judged not only on performance but also on environmental responsibility. Glycol selection now includes lifecycle considerations such as emissions, chemical loss, energy consumption, and recovery efficiency.
MEG systems must minimize discharge losses to prevent environmental contamination. Advanced recovery plants are designed to reclaim nearly all injected glycol, reducing both cost and environmental footprint.
TEG systems focus on energy efficiency. Improved heat integration and lower emissions from regeneration units are becoming standard design priorities.
Both systems are evolving to meet stricter ESG expectations while maintaining operational reliability.
Risk Management and Operational Reliability
From a risk perspective, glycol selection influences operational continuity. Hydrate blockages can shut down entire fields. Poor dehydration can damage pipelines and compressors.
MEG systems reduce catastrophic flow assurance risk. TEG systems ensure long-term infrastructure integrity. The safest operations recognize that gas dehydration is not a single event, but a continuous protection strategy.
Engineers must view glycol choice as part of asset integrity planning, not just chemical selection.
Conclusion: MEG vs TEG Is a Strategic Choice, Not a Chemical Preference
Choosing between MEG and TEG is not about selecting a “better” glycol — it is about selecting the right protection strategy for the operating environment. Each glycol plays a fundamentally different role in gas production systems, and confusing those roles can lead to inefficient designs, higher costs, and increased operational risk.
TEG remains the backbone of gas dehydration where the objective is to deliver pipeline-quality, dry gas. Its ability to achieve very low water content, combined with compact plant design and predictable operation, makes it indispensable in processing facilities. For operators managing centralized gas plants with stable infrastructure, TEG provides consistency, compliance, and long-term reliability.
MEG, on the other hand, is a flow assurance solution first and foremost. It enables gas production in environments where dehydration is impractical or impossible at the point of production. Long subsea tiebacks, deepwater developments, and cold operating conditions all demand hydrate prevention rather than water removal. In these scenarios, MEG allows production to continue safely by stabilizing multiphase flow over extended distances.
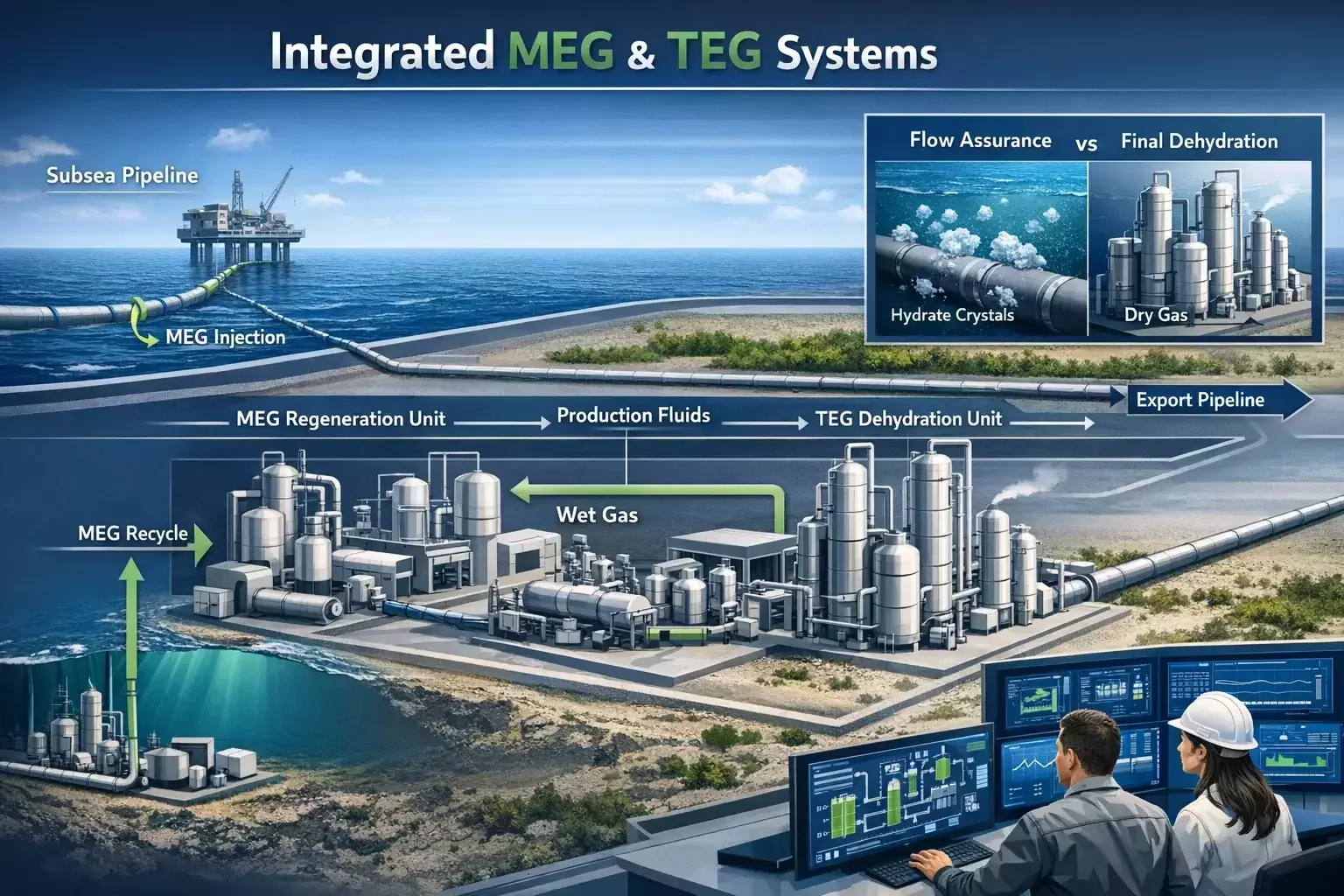
Modern gas developments increasingly rely on both systems working together. MEG protects the flowline and transport network, while TEG completes dehydration at the processing stage. This integrated approach reflects how gas production has evolved — from simple dehydration challenges to complex, system-wide reliability problems.
As the industry moves toward deeper fields, longer tiebacks, and stricter environmental oversight, glycol systems must be designed with a lifecycle mindset. Chemical performance, regeneration efficiency, recovery rates, corrosion control, and emissions management are now interconnected decisions rather than isolated technical choices.
Ultimately, the most successful gas dehydration strategies are those that treat MEG and TEG as complementary tools — applied deliberately, monitored continuously, and optimized as field conditions change.
FAQs
What is the main difference between MEG and TEG in gas systems?
MEG is primarily used to prevent hydrate formation by lowering the freezing point of water in multiphase flow, while TEG is used to remove water vapor from gas to meet pipeline moisture specifications. Their functions address different risks.
Can MEG replace TEG in gas dehydration?
No. MEG does not dehydrate gas to pipeline-quality dryness. It prevents hydrates but leaves water in the system. TEG is required when strict moisture limits must be met before gas export or compression.
Why is MEG preferred in offshore and subsea developments?
Offshore and subsea systems operate at low temperatures and high pressures where hydrate risk is extreme. Dehydration at the wellhead is often impractical, making MEG the most reliable flow assurance solution during transport.
Is TEG suitable for offshore platforms?
Yes, TEG is widely used on offshore platforms where sufficient space, utilities, and regeneration infrastructure are available. It is commonly used downstream of MEG systems in offshore developments.
Which system has higher operating costs?
Costs depend on design and recovery efficiency. MEG systems require large chemical volumes and complex regeneration but reduce hydrate risk. TEG systems consume more energy for regeneration but require less chemical inventory.
Do MEG and TEG systems affect corrosion differently?
Both systems influence corrosion indirectly. MEG travels through aggressive environments and often requires additional corrosion inhibitors. TEG systems are more controlled but require monitoring to prevent degradation-related corrosion.
Can both systems be used in the same field?
Yes. Many modern gas developments use MEG for upstream flow assurance and TEG for downstream dehydration. This combined strategy offers the highest reliability for complex production systems.




