
Introduction: Pipelines as the Lifeline of Oil & Gas Operations
Pipelines form the backbone of the oil and gas industry. From transporting crude oil and natural gas across vast distances to connecting wells, processing facilities, refineries, and export terminals, pipelines ensure uninterrupted energy flow. Yet despite advanced engineering, high-grade metallurgy, and modern monitoring systems, pipelines remain vulnerable to one persistent enemy: corrosion.
Pipeline corrosion is not merely a maintenance issue—it is a major operational, safety, environmental, and financial risk. Corrosion-related failures can lead to leaks, production shutdowns, environmental contamination, regulatory penalties, and in extreme cases, loss of life. Industry studies consistently show that corrosion accounts for a significant percentage of pipeline failures worldwide.
What makes corrosion particularly challenging is that it does not occur in just one way. Pipelines face internal corrosion, driven by the fluids they carry, and external corrosion, caused by environmental exposure. Each form originates from different mechanisms, affects pipelines differently, and requires distinct control strategies.
Understanding the difference between internal and external corrosion is essential for designing effective protection programs. Treating both with the same approach often leads to incomplete protection, higher costs, and unexpected failures.
What Is Pipeline Corrosion? A Practical Explanation
Corrosion is the gradual degradation of metal due to chemical or electrochemical reactions with its surroundings. In oil and gas pipelines, this typically involves steel reacting with water, gases, salts, microorganisms, or soil constituents.
At its core, corrosion is an electrochemical process where metal atoms lose electrons and form corrosion products such as iron oxides, sulfides, or hydroxides. Over time, this process reduces wall thickness, weakens structural integrity, and creates localized pits or cracks.
Unlike uniform wear, corrosion is often localized and unpredictable. A pipeline may appear intact externally while severe internal pitting progresses unnoticed. Similarly, coatings may mask external corrosion until significant damage has already occurred.
This hidden nature is what makes corrosion management both technically complex and operationally critical.
Why Corrosion Control Is a Strategic Priority in Oil & Gas
Pipeline corrosion affects every segment of the oil and gas value chain:
Upstream: Flowlines carrying multiphase fluids with water, CO₂, H₂S, and solids
Midstream: Transmission pipelines exposed to soil, groundwater, and varying operating conditions
Downstream: Refinery piping systems handling corrosive products, acids, and high temperatures
As fields mature, corrosion risks often increase rather than decrease. Rising water cuts, changing fluid chemistry, and aging infrastructure all accelerate corrosion mechanisms.
Regulators worldwide now require operators to demonstrate proactive corrosion management programs. Beyond compliance, companies increasingly recognize that effective corrosion control directly impacts asset life, operating costs, and corporate reputation.
Internal vs External Corrosion: Two Distinct Threats
Although both forms damage pipelines, internal and external corrosion differ fundamentally in how they originate and how they must be controlled.
Internal Corrosion: The Threat from Within
Internal corrosion occurs on the inside surface of the pipeline and is driven by the characteristics of the transported fluid. It is typically influenced by:
Presence of water (free water or condensation)
Acidic gases such as carbon dioxide (CO₂) and hydrogen sulfide (H₂S)
Dissolved oxygen
Salts and organic acids
Microbial activity, especially sulfate-reducing bacteria
Internal corrosion is particularly dangerous because it often develops out of sight. Without proper monitoring and chemical treatment, metal loss can progress rapidly and remain undetected until failure occurs.
External Corrosion: Environmental Attack from the Outside
External corrosion occurs when the outer surface of the pipeline interacts with its surrounding environment. This can include:
Soil moisture and chemistry
Groundwater salinity
Atmospheric exposure in above-ground pipelines
Mechanical damage to protective coatings
Stray electrical currents
Unlike internal corrosion, external corrosion is heavily influenced by location, soil conditions, climate, and the effectiveness of protective coatings and cathodic protection systems.
Why Treating Them Separately Matters
One of the most common mistakes in pipeline integrity management is assuming that corrosion control is a single problem with a single solution. In reality, internal and external corrosion require separate assessment, monitoring, and mitigation strategies.
A pipeline with excellent external coating and cathodic protection can still fail due to severe internal corrosion. Conversely, a well-treated internal fluid system offers no protection against soil-induced corrosion if coatings or cathodic systems fail.
Effective corrosion control begins with understanding which mechanisms are active, where they occur, and how they interact over time.
The Cost of Inadequate Corrosion Management
Failure to properly distinguish and control internal and external corrosion leads to:
Unexpected pipeline leaks and ruptures
Emergency shutdowns and production losses
Costly repairs and replacements
Environmental damage and cleanup liabilities
Regulatory penalties and reputational harm
Proactive corrosion control is far more cost-effective than reactive repair. This is why modern operators invest heavily in corrosion monitoring, chemical treatment programs, and integrity management systems.
Understanding Internal Corrosion from the Inside Out
Internal corrosion develops on the inner surface of pipelines and is directly influenced by the nature of the fluids being transported. In oil and gas systems, pipelines rarely carry “dry” hydrocarbons. Instead, they transport complex mixtures of oil, gas, water, dissolved gases, solids, and treatment chemicals. This internal environment creates multiple corrosion-driving conditions that can act simultaneously.
What makes internal corrosion particularly challenging is that it evolves continuously as reservoir conditions change. Water cut increases over time, gas composition fluctuates, and operating pressures and temperatures vary—each factor influencing corrosion behavior in different ways.

The Role of Water: The Primary Corrosion Enabler
Water is the single most important factor in internal corrosion. Dry hydrocarbons alone are generally non-corrosive, but once water is present, corrosion mechanisms become active.
In pipelines, water can appear in several forms:
Free water separated from hydrocarbons
Condensed water from wet gas systems
Produced water containing salts and organic acids
Injection water entering production lines
Water acts as an electrolyte, enabling electrochemical reactions between steel and corrosive species. As water content increases, corrosion rates typically rise—especially when water becomes continuous rather than dispersed.
Carbon Dioxide (CO₂) Corrosion – “Sweet Corrosion”
Carbon dioxide is one of the most common corrosive gases in oil and gas production. When CO₂ dissolves in water, it forms carbonic acid, lowering pH and accelerating metal dissolution.
CO₂ corrosion often results in:
Uniform wall thinning
Localized pitting under certain flow conditions
Formation of iron carbonate scales, which may or may not be protective
While some iron carbonate layers can slow corrosion, they are unstable under changing flow rates, temperature shifts, or mechanical disturbance. Once disrupted, corrosion can accelerate rapidly.
Hydrogen Sulfide (H₂S) and Sour Corrosion
Hydrogen sulfide introduces a more aggressive corrosion environment. When dissolved in water, H₂S forms weak acids and reacts with iron to produce iron sulfide scales.
Although some sulfide films appear protective, they often conceal severe localized corrosion underneath. H₂S corrosion is particularly dangerous because it:
Promotes pitting and cracking
Increases the risk of sulfide stress cracking (SSC)
Creates safety hazards due to toxic gas release
Sour systems require corrosion strategies that address both chemical attack and mechanical integrity.
Microbiologically Influenced Corrosion (MIC)
Internal corrosion is not always purely chemical. Microorganisms—especially sulfate-reducing bacteria—can dramatically accelerate metal loss.
MIC occurs when bacteria:
Form biofilms on pipe walls
Produce corrosive by-products such as organic acids and hydrogen sulfide
Create localized electrochemical cells beneath biofilms
This type of corrosion is highly localized and often severe, leading to unexpected failures even in pipelines with relatively short service life. MIC is particularly common in low-flow or stagnant areas such as dead legs, low points, and separators.
Flow Regime and Corrosion Interaction
Internal corrosion is strongly influenced by how fluids move through the pipeline. Turbulent flow, slug flow, and stratified flow all affect corrosion behavior differently.
High flow velocities may remove protective corrosion films, increasing metal exposure. Low velocities allow water and solids to settle, creating ideal conditions for localized corrosion and microbial growth.
Multiphase flow adds another layer of complexity, as alternating contact between gas, oil, and water can repeatedly disrupt protective layers and expose fresh metal surfaces.
Internal Corrosion Monitoring Challenges
Detecting internal corrosion is inherently difficult. Unlike external corrosion, it cannot be visually inspected without interrupting operations.
Operators rely on a combination of:
Corrosion probes and coupons
Inline inspection tools (smart pigs)
Fluid sampling and water chemistry analysis
Microbial monitoring
Even with these tools, corrosion can develop between inspection intervals, making proactive control essential.
Chemical Control Strategies for Internal Corrosion
Chemical treatment remains the most effective method for managing internal corrosion in operating pipelines. These strategies are designed to either prevent corrosive reactions or mitigate their impact.
Corrosion Inhibitors
Film-forming corrosion inhibitors are widely used to protect internal surfaces. These chemicals adsorb onto the metal surface, creating a barrier that limits contact between steel and corrosive fluids.
Proper inhibitor selection depends on:
Fluid composition
Temperature and pressure
Flow regime
Presence of CO₂, H₂S, and solids
Consistent dosing and monitoring are critical to ensure continuous protection.
Biocides
Biocides play a vital role in controlling MIC. By limiting microbial populations, biocides reduce biofilm formation and the production of corrosive metabolites.
Effective programs often combine periodic shock dosing with maintenance treatments to prevent bacterial adaptation.
pH Control and Oxygen Scavenging
Adjusting fluid pH and removing dissolved oxygen can significantly reduce corrosion rates. Oxygen scavengers are particularly important in systems where oxygen ingress is possible, such as water injection pipelines.
Why Internal Corrosion Demands Continuous Attention
Internal corrosion is not a one-time problem that can be “fixed” and forgotten. As production conditions evolve, corrosion risks change accordingly.
A pipeline that operates safely for years may suddenly experience accelerated corrosion due to:
Increased water production
Changes in gas composition
Altered flow conditions
Inadequate chemical dosing
This dynamic nature makes internal corrosion management a continuous process rather than a static solution.
Transition to External Corrosion Considerations
While internal corrosion attacks from within, pipelines simultaneously face threats from their external environment. Understanding internal corrosion sets the foundation for appreciating why external corrosion requires entirely different protection strategies.
In the next section, we will explore external corrosion in oil & gas pipelines, focusing on environmental factors, coating systems, and cathodic protection methods that safeguard pipelines from the outside.What Is External Corrosion and Why It’s a Persistent Threat
External corrosion occurs on the outer surface of pipelines and is driven by the environment surrounding the asset rather than the fluids flowing inside it. While internal corrosion is influenced by process conditions, external corrosion is governed by soil chemistry, moisture, atmospheric exposure, and mechanical damage to protective systems.
Unlike internal corrosion, which is often managed through chemical dosing, external corrosion control relies heavily on engineering design, coatings, and electrochemical protection. However, once these systems are compromised, corrosion can progress unnoticed for years until a failure occurs.
This makes external corrosion particularly dangerous for buried pipelines, offshore subsea lines, and long-distance transmission networks where inspection access is limited.
Soil as a Corrosive Medium
For buried pipelines, soil is not a passive environment. It acts as an electrolyte that enables corrosion reactions, especially when moisture and dissolved salts are present.
Several soil-related factors influence corrosion severity:
Soil resistivity, which determines how easily electrical currents flow
Moisture content, which activates electrochemical reactions
Chlorides and sulfates, which accelerate metal dissolution
Soil pH, where acidic conditions increase corrosion rates
Presence of stray electrical currents from nearby infrastructure
Low-resistivity soils with high moisture and salt content are particularly aggressive, creating ideal conditions for sustained corrosion activity along the pipeline surface.
Atmospheric Corrosion in Above-Ground Pipelines
Pipelines exposed to the atmosphere face a different set of challenges. Atmospheric corrosion is driven by oxygen, humidity, temperature cycling, and airborne contaminants.
Above-ground pipelines in coastal, industrial, or desert environments experience:
Salt deposition from marine aerosols
Sulfur compounds from industrial emissions
Condensation cycles caused by temperature fluctuations
UV degradation of protective coatings
These conditions can cause coating breakdown, exposing bare metal to continuous corrosion attack. Even small coating defects can grow into widespread corrosion zones over time.
External Corrosion in Offshore and Subsea Environments
Offshore pipelines operate in one of the most corrosive environments on earth. Seawater is highly conductive and rich in chlorides, making corrosion reactions extremely efficient.
Subsea pipelines face additional challenges:
Continuous immersion in seawater
Microbial activity in seabed sediments
Mechanical damage during installation
Differential oxygen concentrations along the pipe length
In shallow waters, wave action and tidal effects further stress coatings and protective layers. In deepwater systems, high pressure and low temperature add complexity to corrosion protection design.
The Critical Role of Protective Coatings
Protective coatings form the first line of defense against external corrosion. Their primary function is to physically isolate the pipeline surface from the surrounding environment.
Common pipeline coating systems include:
Fusion-bonded epoxy (FBE)
Three-layer polyethylene or polypropylene systems
Coal tar enamel (legacy systems)
Liquid-applied epoxy and polyurethane coatings
A well-applied coating significantly reduces corrosion risk, but coatings are not permanent. Mechanical damage during handling, installation, or ground movement can create defects that allow localized corrosion to initiate.
Once corrosion starts beneath a coating defect, it often spreads unseen, making early detection difficult.
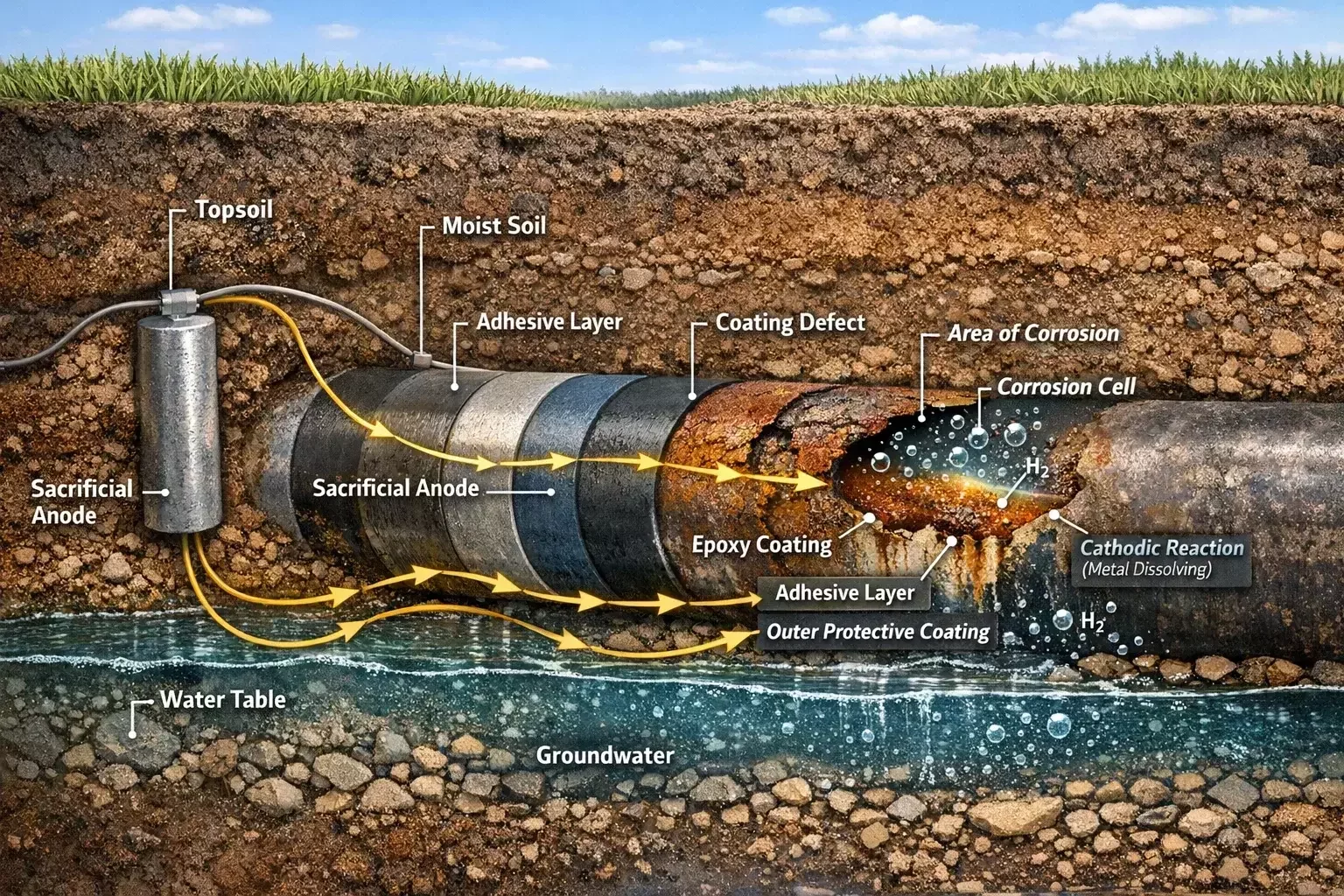
Cathodic Protection: The Backbone of External Corrosion Control
Because coatings alone cannot guarantee long-term protection, cathodic protection (CP) systems are used as a secondary defense.
Cathodic protection works by shifting the electrochemical potential of the pipeline so that corrosion reactions are suppressed. This is achieved by making the pipeline the cathode of an electrochemical cell.
Two primary CP methods are used:
Sacrificial anode systems, where reactive metals corrode instead of the pipeline
Impressed current systems, where an external power source provides protective current
When properly designed and maintained, cathodic protection can dramatically extend pipeline life—even in aggressive environments.
Interdependence of Coatings and Cathodic Protection
Coatings and cathodic protection do not function independently. They are designed to work together.
Coatings reduce the surface area requiring protection, allowing cathodic protection systems to operate efficiently. Conversely, cathodic protection compensates for coating defects by preventing corrosion at exposed areas.
If either system fails, the burden on the other increases. Poor coatings demand higher CP current, while inadequate CP allows corrosion to initiate at coating flaws.
Stray Current Corrosion Risks
External corrosion can also be driven by stray electrical currents originating from nearby infrastructure such as railways, power lines, or industrial facilities.
Stray current corrosion is particularly dangerous because it can cause rapid, localized metal loss. Pipelines located near electrified rail systems or DC power installations are especially vulnerable.
Mitigating stray current corrosion requires specialized grounding, insulation joints, and continuous monitoring to ensure protective systems remain effective.
Inspection and Monitoring of External Corrosion
External corrosion is typically monitored through indirect inspection techniques rather than direct visual assessment.
Common monitoring approaches include:
Cathodic protection potential surveys
Close-interval potential surveys (CIPS)
Direct current voltage gradient (DCVG) surveys
Coating integrity assessments
Excavation and direct examination at high-risk locations
These methods help operators identify coating damage, CP deficiencies, and corrosion hotspots before failures occur.
Why External Corrosion Failures Are Often Sudden
One of the most dangerous aspects of external corrosion is its ability to remain undetected for long periods. Corrosion beneath coatings or in buried sections can progress silently until the remaining wall thickness is insufficient to withstand operating pressure.
When failure occurs, it is often sudden and severe, leading to:
Environmental damage
Safety incidents
Regulatory penalties
Costly downtime and repairs
This makes proactive external corrosion management essential for pipeline integrity.
Setting the Stage for Integrated Corrosion Control
External corrosion cannot be managed in isolation. Pipelines are simultaneously exposed to internal and external threats, each requiring different control strategies.
Understanding external corrosion highlights why pipeline integrity programs must integrate chemical treatment, engineering design, inspection, and monitoring into a unified approach.
In the final section, we will bring these perspectives together by comparing internal vs external corrosion control strategies and exploring how integrated programs deliver long-term reliability and cost efficiency.Why Corrosion Control Cannot Be Treated in Isolation
In real-world oil and gas operations, pipelines are exposed to both internal and external corrosion risks at the same time. Treating these threats as separate challenges often leads to gaps in protection, duplicated costs, or unexpected failures.
Internal corrosion may weaken the pipe wall from the inside due to corrosive fluids, while external corrosion attacks from the outside through soil, water, or atmospheric exposure. When both processes act simultaneously, the combined metal loss can significantly shorten pipeline life—even when each corrosion mechanism appears manageable on its own.
This is why modern pipeline integrity programs focus on integrated corrosion control strategies rather than isolated solutions.
Comparing Internal and External Corrosion Control Approaches
Internal corrosion control is primarily chemical-driven. It relies on continuous monitoring of fluid composition and targeted chemical treatment programs. Corrosion inhibitors, biocides, oxygen scavengers, and scale inhibitors are adjusted based on operating conditions, production chemistry, and monitoring data.
External corrosion control, by contrast, is engineering-driven. It depends on physical barriers such as coatings, electrochemical systems like cathodic protection, and environmental monitoring. Once installed, these systems require regular verification rather than constant adjustment.
Because the tools, expertise, and monitoring techniques differ, it is easy for organizations to manage them through separate teams. However, this separation often creates blind spots where corrosion risks go unnoticed.
How Integrated Corrosion Management Improves Reliability
An integrated corrosion management program aligns internal and external strategies under a single integrity framework. Instead of reacting to corrosion events, operators focus on preventing them through coordinated planning and data sharing.
For example, internal corrosion data showing elevated water cut or microbial activity can signal increased risk at low points where external corrosion may also accelerate. Similarly, external inspection results identifying coating damage can prompt internal chemistry reviews to ensure adequate inhibitor protection.
By linking internal chemistry trends with external condition monitoring, operators gain a more accurate understanding of actual pipeline health.

The Role of Data and Monitoring in Integration
Modern corrosion control increasingly relies on data-driven decision-making. Inline inspection results, corrosion coupons, electrical resistance probes, cathodic protection surveys, and fluid analysis data are no longer viewed independently.
When these data streams are combined, they reveal patterns that would otherwise remain hidden. Corrosion rate spikes, pressure fluctuations, temperature changes, and chemical consumption trends can be correlated to identify emerging risks early.
This integrated visibility allows operators to move from reactive maintenance to predictive integrity management.
Balancing Cost, Risk, and Performance
One of the biggest challenges in corrosion control is balancing protection with cost efficiency. Over-treatment increases chemical and operational expenses, while under-treatment increases failure risk.
Integrated corrosion control helps optimize spending by:
Targeting inhibitors only where internal corrosion risk exists
Adjusting cathodic protection based on coating condition
Reducing unnecessary chemical dosing through better diagnostics
Prioritizing inspections in high-risk pipeline segments
This targeted approach ensures resources are spent where they deliver the highest risk reduction.
Regulatory and Environmental Implications
Regulatory expectations increasingly demand evidence of systematic pipeline integrity management. Authorities no longer accept reactive repairs as proof of compliance.
Integrated corrosion control supports compliance by demonstrating:
Proactive risk identification
Continuous monitoring and documentation
Preventive maintenance strategies
Reduced likelihood of environmental incidents
From an environmental perspective, preventing corrosion-related leaks is far more effective than responding after a failure occurs. Integrated programs align well with sustainability and ESG objectives by minimizing spill risk and asset loss.
The Importance of Expertise and Partnership
Corrosion control is not a one-size-fits-all solution. Each pipeline system has unique operating conditions, fluid compositions, environmental exposure, and lifecycle considerations.
Effective integration requires collaboration between:
Production and process engineers
Corrosion specialists
Chemical suppliers
Inspection and integrity teams
Suppliers with broad chemical and technical expertise play a key role in designing programs that address both internal and external risks without conflict or redundancy.
Conclusion: Internal vs External Corrosion Control Is Not a Choice — It’s a Combination
Internal and external corrosion are fundamentally different in how they occur, how they are controlled, and how they are monitored. Treating them as separate challenges is a legacy approach that no longer meets the demands of modern oil and gas operations.
Internal corrosion control protects pipelines from aggressive fluids, microbial activity, and chemical reactions occurring inside the system. External corrosion control shields assets from soil, water, atmosphere, and electrical influences acting from the outside.
Long-term pipeline reliability depends on how well these two strategies are integrated. When chemical treatment programs, engineering systems, inspection data, and operational insights are aligned, operators gain a holistic view of pipeline health.
This integrated approach reduces failures, extends asset life, improves safety, and supports regulatory and environmental responsibilities. In today’s complex operating environments, effective corrosion control is not about choosing between internal or external protection—it is about managing both together, intelligently and continuously.
FAQs
What is the main difference between internal and external pipeline corrosion?
Internal corrosion is caused by fluids flowing inside the pipeline, such as water, CO₂, H₂S, and microbes. External corrosion is driven by environmental exposure, including soil, seawater, and atmospheric conditions.
Can corrosion inhibitors protect against external corrosion?
No. Corrosion inhibitors are designed for internal protection. External corrosion is controlled through coatings, cathodic protection, and environmental management.
Why do pipelines fail even with corrosion protection systems in place?
Failures often occur due to system gaps—such as coating damage without adequate cathodic protection, or internal corrosion progressing unnoticed due to insufficient monitoring.
How often should corrosion control systems be reviewed?
Both internal and external systems should be reviewed continuously using monitoring data, with formal assessments conducted at defined intervals based on risk and regulatory requirements.
Is integrated corrosion control more expensive?
While integration requires planning and coordination, it often reduces long-term costs by preventing failures, optimizing chemical use, and extending asset life.




