Introduction
Scale deposition is one of the most persistent and costly flow assurance challenges in oil and gas production. Whether in upstream production tubing, surface facilities, or injection systems, mineral scale reduces permeability, restricts fluid flow, damages equipment, and increases operational downtime. Over time, even a thin layer of scale can significantly impact heat transfer efficiency, pumping energy, and overall production output. Operators worldwide invest millions annually in scale prevention and remediation because uncontrolled scaling directly translates into lost revenue and increased maintenance costs.
Among the advanced chemical solutions used for scale removal, chelating agents — particularly EDTA (Ethylenediaminetetraacetic acid) and DTPA (Diethylenetriaminepentaacetic acid) — have emerged as highly effective alternatives to conventional acid treatments. These compounds are not just dissolvers; they function as targeted molecular binders that safely capture and remove metal ions responsible for scale formation. Their ability to dissolve stubborn inorganic deposits without aggressively attacking base metals makes them indispensable in modern oilfield chemistry.
This article explores the chemistry, mechanism, operational benefits, and field applications of EDTA and DTPA in oil and gas scale removal, providing a technical yet practical understanding of why chelating agents are becoming increasingly preferred in complex production environments.

Understanding Scale Formation in Oil & Gas Systems
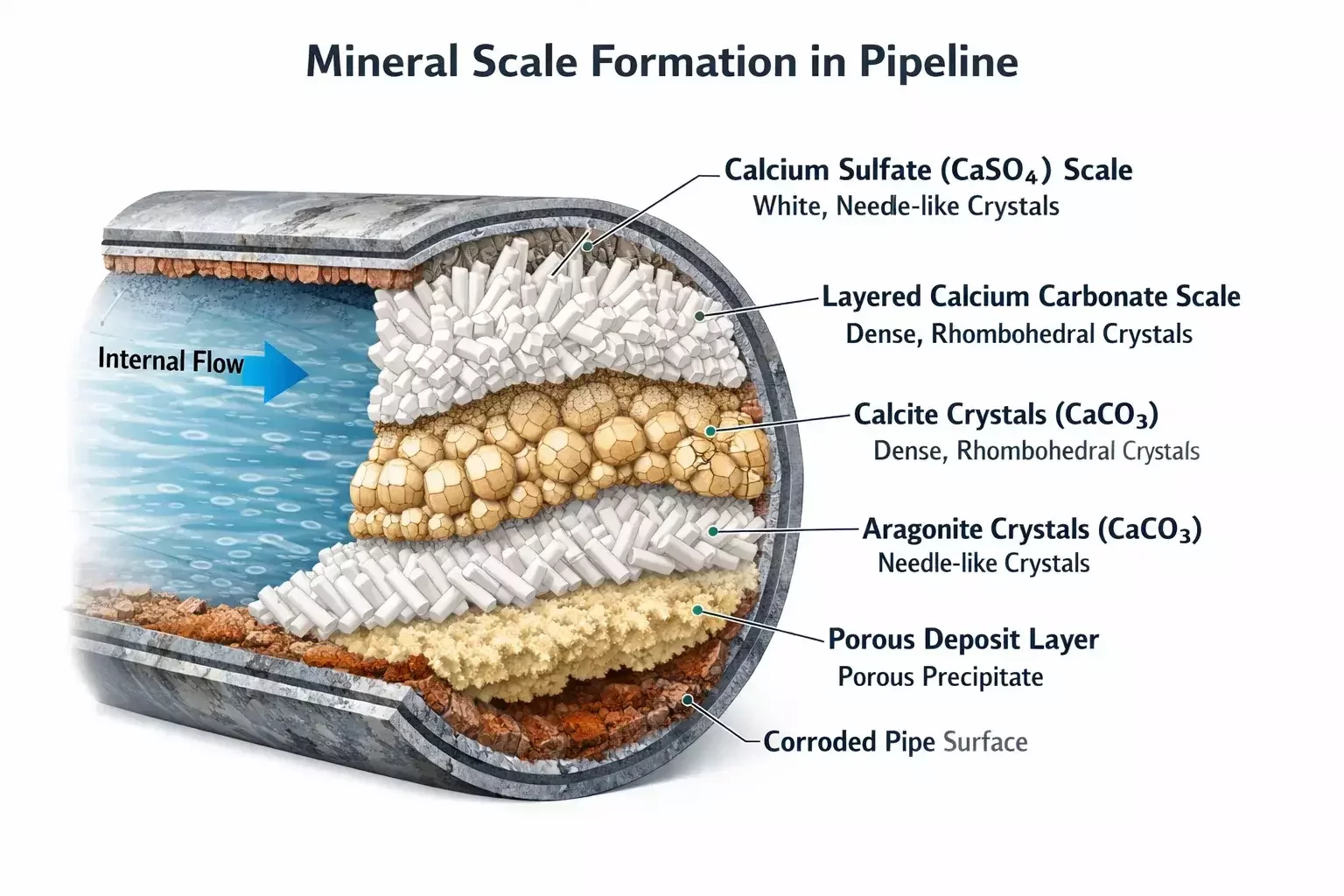
Scale formation occurs when dissolved mineral salts in produced water become supersaturated and precipitate onto surfaces. The process is influenced by pressure changes, temperature variations, fluid mixing, and chemical imbalance. When formation water, injection water, or seawater interact under production conditions, incompatible ions combine and crystallize, forming hard mineral deposits.
The most common oilfield scales include calcium carbonate, calcium sulfate, barium sulfate, and strontium sulfate. Carbonate scales typically form due to pressure drops that release dissolved carbon dioxide, shifting pH and promoting precipitation. Sulfate scales, on the other hand, often result from mixing incompatible waters — such as sulfate-rich seawater with barium-rich formation brine. These scales are particularly problematic because they are dense, adherent, and resistant to mechanical removal.
Traditional acid treatments like hydrochloric acid are effective against carbonate scales but have limitations. Strong acids can corrode tubulars, damage elastomers, and create safety hazards during handling. Moreover, sulfate scales are largely acid-insoluble, making them resistant to conventional treatments. This is where chelating chemistry becomes essential. Instead of relying on aggressive dissolution, chelating agents selectively bind metal ions and convert solid scale into soluble complexes.
The advantage of this approach is precision. Rather than attacking the entire system chemically, chelating agents target the root cause — the metal ions themselves — allowing for controlled and safer scale removal.
What Are Chelating Agents?
Chelating agents are specialized organic molecules designed to form stable, water-soluble complexes with metal ions. The term “chelate” originates from the Greek word for claw, which reflects how these molecules wrap around metal ions and hold them in a secure molecular grip. Once bound, the metal ions remain suspended in solution, preventing re-precipitation.
EDTA and DTPA are polyaminocarboxylic acids with multiple binding sites capable of attaching to divalent and trivalent metal ions such as calcium, magnesium, iron, and barium. Their structure allows them to function like molecular cages. This cage-like binding is extremely stable, which is why these chelants are widely used not only in oilfields but also in water treatment, medicine, and industrial cleaning.
In oil and gas operations, chelating agents are formulated into scale dissolvers that penetrate deposits and gradually break down crystalline structures. Unlike acids, chelants operate effectively across a wider pH range and generate minimal heat during reaction. This controlled reaction profile reduces risk to equipment and personnel while maintaining high dissolution efficiency.
Another critical benefit is compatibility. Chelant-based systems can be engineered to work alongside corrosion inhibitors, surfactants, and other oilfield chemicals. This flexibility allows operators to tailor treatment packages for specific reservoirs and facility conditions without compromising system integrity.
Why EDTA and DTPA Are Preferred in Oilfield Applications
Although many chelating agents exist, EDTA and DTPA dominate oilfield scale removal due to their balance of strength, stability, and field reliability. EDTA is highly effective against calcium and iron-based scales and performs well in moderate temperature environments. DTPA, with its additional binding groups, provides enhanced stability at higher temperatures and stronger affinity for difficult scales such as barium sulfate.
In high-temperature wells where conventional chemicals degrade, DTPA maintains performance and continues to solubilize scale efficiently. This thermal stability is critical for deep wells and enhanced recovery operations. Furthermore, both EDTA and DTPA reduce the risk of secondary precipitation — a common issue when poorly designed treatments redeposit dissolved minerals elsewhere in the system.
Their slower reaction kinetics compared to strong acids also offer operational advantages. The treatment can penetrate deeper into scale layers, ensuring more uniform dissolution instead of surface-only removal. This leads to longer-lasting results and improved production restoration.
Mechanism of Chelation in Scale Dissolution
Chelation-driven scale removal is fundamentally different from acid dissolution because it is based on selective molecular interaction rather than aggressive chemical attack. When a chelating agent such as EDTA or DTPA is introduced into a scaled system, it does not simply dissolve the surface layer. Instead, it diffuses into microscopic fractures and pores within the scale structure. Once inside, the chelant molecules begin binding with metal ions that hold the crystal lattice together.
Scale deposits are stabilized by ionic bonds between metal cations and anions. Chelating agents disrupt these bonds by forming stronger coordination complexes with the metal ions. Because the chelant-metal complex is thermodynamically more stable than the original scale compound, the solid mineral structure collapses and transitions into a soluble form. This process continues progressively, dissolving the scale from within rather than eroding it externally.
The kinetics of chelation are influenced by temperature, pH, chelant concentration, and fluid circulation rate. Higher temperatures generally accelerate reaction rates, which is why DTPA is often preferred in high-temperature reservoirs. pH control is equally important. Chelating agents perform optimally in slightly alkaline environments where their binding sites remain active. Improper pH can reduce efficiency or lead to incomplete dissolution.
Another important factor is iron control. During scale removal, iron ions released from deposits or corrosion can interfere with treatment performance. EDTA and DTPA exhibit strong iron-binding capacity, preventing iron precipitation and maintaining fluid clarity. This dual function — scale removal and iron stabilization — is one of the reasons chelants are favored in mature fields with corrosion history.
Unlike acid reactions that produce gas and heat, chelation is a relatively calm process. The absence of violent reaction reduces risk of formation damage, tubing stress, or sudden pressure spikes. For sensitive completions and aging infrastructure, this controlled chemistry is a major operational advantage.

Field Applications of EDTA and DTPA Treatments
Chelant-based scale removal is widely applied across upstream, midstream, and surface production facilities. In downhole environments, EDTA and DTPA treatments are commonly used to restore productivity in wells affected by carbonate or sulfate scaling. These treatments are delivered through bullheading, coiled tubing placement, or circulation systems depending on well configuration.
In production tubing, scale buildup narrows flow paths and increases frictional pressure losses. Chelant treatments dissolve these deposits without aggressive corrosion, preserving tubing integrity. This is particularly important in older wells where metal fatigue and corrosion risks are already elevated.
Surface facilities such as separators, heat exchangers, and injection lines also benefit from chelant cleaning. Heat exchangers, for example, are highly sensitive to scale because even thin deposits drastically reduce thermal efficiency. Chelant cleaning solutions circulate through the equipment, dissolving deposits without requiring dismantling. This minimizes shutdown time and reduces maintenance costs.
In water injection systems, scale formation can impair injectivity and damage pumps. Chelant treatments remove deposits while maintaining compatibility with injection water chemistry. Operators often integrate chelant cleaning into routine maintenance schedules to prevent catastrophic buildup.
Another emerging application is in enhanced oil recovery (EOR) systems, where chemical flooding and water management create complex scaling risks. Chelants provide targeted remediation without interfering with polymer or surfactant performance, making them valuable in integrated chemical programs.
Operational Advantages Over Conventional Acid Systems
The shift from acid-based scale removal to chelant systems is driven by safety, efficiency, and asset protection. Strong mineral acids such as hydrochloric acid react rapidly and aggressively. While effective against carbonate scales, they pose serious corrosion hazards and require heavy inhibitor loading. Even with inhibitors, acid exposure shortens equipment lifespan and increases maintenance frequency.
Chelating agents operate with far lower corrosion risk. Because they bind selectively with metal ions rather than indiscriminately attacking surfaces, they are gentler on tubulars and completion hardware. This allows longer contact time, deeper penetration, and more thorough cleaning.
Another operational benefit is compatibility with complex metallurgy. Modern wells contain alloys, elastomers, and sensitive components that may not tolerate acid exposure. Chelant systems can be formulated to remain safe across mixed materials, reducing risk of equipment failure.
Logistics and safety also improve with chelant treatments. They are easier to transport, store, and handle compared to concentrated acids. The reduced hazard profile lowers operational risk and simplifies regulatory compliance. In offshore or remote environments where safety margins are critical, this advantage becomes even more significant.
Chelant treatments also generate fewer secondary problems. Acid reactions can produce sludge, precipitates, or emulsions that require additional cleanup. Chelants maintain dissolved metals in solution, minimizing the chance of redeposition elsewhere in the system.
Environmental and Safety Considerations
Modern oil and gas operations operate under increasing environmental scrutiny, and chemical treatments are evaluated not only for performance but also for ecological footprint. Chelating agents such as EDTA and DTPA are often viewed as safer alternatives to strong mineral acids because they reduce corrosion risk, minimize hazardous fumes, and lower handling danger during transport and mixing. However, responsible deployment still requires careful design and regulatory awareness.
One environmental consideration is persistence. Chelants are highly stable molecules by design — their ability to tightly bind metals is what makes them effective. That same stability means they can remain active in produced water streams if not properly neutralized or treated. Many operators integrate post-treatment water management strategies to ensure chelant-metal complexes are processed safely before discharge or reinjection.
Advances in biodegradable chelant formulations are helping address this challenge. Modified EDTA/DTPA derivatives and environmentally optimized chelant blends are increasingly used in regions with strict discharge regulations. These formulations maintain performance while improving breakdown rates under controlled conditions.
From a safety standpoint, chelant treatments significantly reduce the acute hazards associated with acid stimulation. They do not produce corrosive vapors, violent exothermic reactions, or sudden gas evolution. This calmer reaction profile improves worker safety, reduces PPE burden, and lowers the risk of accidental exposure. In offshore or high-pressure facilities, where emergency response is complex, these safety improvements are operationally meaningful.
Ultimately, environmental performance depends less on the chemical itself and more on the treatment program design. Proper dosage, containment, recovery, and disposal practices ensure chelant systems remain aligned with sustainability goals.
Treatment Design and Optimization Strategies
Successful chelant-based scale removal is not simply a matter of injecting a chemical solution. Effective treatment requires a carefully engineered program tailored to scale composition, reservoir conditions, and operational objectives. Pre-treatment diagnostics are essential. Scale samples are typically analyzed using X-ray diffraction, spectroscopy, or ion analysis to determine mineral composition and metal content. This information guides chelant selection and concentration.
Temperature plays a central role in treatment design. High-temperature wells favor DTPA due to its thermal stability, while EDTA remains effective in moderate environments. Contact time is equally critical. Chelation is a diffusion-driven process, so sufficient soak time must be allowed for deep penetration into scale deposits. Circulation patterns are engineered to maximize fluid contact without causing formation damage.
pH buffering is another optimization factor. Chelants require specific pH ranges to maintain binding efficiency. Buffer systems stabilize the treatment fluid, preventing premature neutralization or performance loss. Corrosion inhibitors, surfactants, and dispersants may also be added to improve fluid placement and metal protection.
In complex wells, staged treatments are sometimes used. A preflush removes loose debris and prepares the surface, followed by chelant placement and a post-flush that clears dissolved scale. This layered approach ensures maximum effectiveness and prevents partial cleaning that could lead to re-scaling.
Field monitoring completes the optimization cycle. Return fluid analysis confirms metal removal efficiency and helps engineers refine future treatments. Over time, data-driven programs reduce chemical consumption while improving production recovery.
Practical Field Insights and Performance Outcomes
Field experience consistently shows that chelant treatments provide longer-lasting results compared to rapid acid washes. Because chelation penetrates deeper into the scale structure, it reduces the likelihood of residual seed crystals that trigger immediate re-precipitation. Operators frequently observe improved flow rates, stabilized pressure profiles, and extended intervals between maintenance interventions.
In mature oilfields where repeated acid treatments have weakened infrastructure, chelants offer a restorative alternative. Wells previously considered high-risk for acidizing can be treated safely with chelant systems, extending asset life without compromising production targets.
Another notable benefit is predictability. Acid reactions can be difficult to control, especially in heterogeneous formations. Chelant treatments follow more stable kinetics, allowing engineers to model outcomes with greater accuracy. This predictability reduces operational uncertainty and improves planning efficiency.
The economics are equally compelling. While chelant systems may carry higher upfront chemical cost, they often reduce total lifecycle expenses by minimizing downtime, corrosion damage, and repeat interventions. When evaluated across the full operational timeline, chelant programs frequently deliver superior return on investment.
Future Trends in Chelant Technology
The oil and gas industry continues to evolve toward smarter, safer, and more sustainable chemistry. Research is advancing next-generation chelants that combine high binding efficiency with enhanced biodegradability. Nanotechnology-assisted delivery systems are being explored to improve penetration into ultra-tight scale matrices. Hybrid formulations that merge chelation with targeted dissolution catalysts are also under development.
Digital modeling and AI-assisted treatment design are emerging as powerful tools. By integrating scale prediction software with chemical performance data, operators can simulate treatments before field deployment. This predictive capability reduces trial-and-error approaches and supports precision chemical engineering.
As production environments grow more complex — including high-pressure, high-temperature reservoirs and unconventional fields — chelant chemistry will likely become even more central to flow assurance strategies. The demand for non-damaging, controllable, and environmentally responsible scale removal solutions aligns directly with the strengths of EDTA and DTPA systems.

Conclusion
Scale removal in oil and gas operations is no longer just a maintenance task; it is a strategic component of production optimization. Chelating agents such as EDTA and DTPA represent a shift from aggressive chemical attack toward precision molecular engineering. By targeting metal ions at the root of scale formation, these agents dissolve deposits safely, effectively, and predictably.
Their advantages extend beyond cleaning power. Reduced corrosion risk, improved safety, compatibility with modern materials, and adaptability to high-temperature environments make chelant systems indispensable in today’s oilfield chemistry toolkit. When supported by proper diagnostics and treatment design, EDTA and DTPA deliver durable performance that protects both assets and production.
As regulatory expectations tighten and operational complexity increases, the industry’s reliance on intelligent chemical solutions will only grow. Chelation technology stands at the intersection of performance and responsibility — offering a pathway to cleaner, safer, and more efficient scale management in the evolving energy landscape.




